文/Sobereva@北京科音
First release: 2024-Feb-16 Last update: 2024-Feb-17
0 前言
Hirshfeld surface分析(以下簡稱HS分析)是展現分子晶體中一個或多個分子與周圍分子間相互作用的方法,它也同樣可以用于展現孤立體系中特定片段與周圍原子間的相互作用。HS分析的原理容易理解,圖像比較直觀,在分子晶體的研究領域已經用得非常普遍。Multiwfn的主功能12中很早以前就已經實現了HS分析,如今已經被不少文章使用。本文將結合許多例子,專門詳細具體講解一下Multiwfn做HS分析的各方面細節、操作和技巧。
本文的讀者請務必使用2024-Feb-16及以后更新的Multiwfn版本,否則與本文所述情況會有很多不同。Multiwfn可以在其主頁http://www.shanxitv.org/multiwfn免費下載。不了解Multiwfn者請參看《Multiwfn FAQ》(http://www.shanxitv.org/452)和《Multiwfn入門tips》(http://www.shanxitv.org/167)。使用Multiwfn做HS分析在寫文章時請按照Multiwfn啟動時的提示對程序進行恰當的引用。
很值得一提的是,筆者提出的IGMH方法也非常適合展現分子晶體中的分子間相互作用,HS分析與IGMH分析的展現形式有明顯區別且有一定程度的互補性,二者亦可以同時使用以提供更多視角。請閱讀《使用Multiwfn做IGMH分析非常清晰直觀地展現化學體系中的相互作用》(http://www.shanxitv.org/621)和《一篇最全面介紹各種弱相互作用可視化分析方法的文章已發表!》(http://www.shanxitv.org/667)提到的綜述了解IGMH的相關知識,里面也專門有IGMH用于分子晶體的實際例子。
1 Hirshfeld surface分析的基本思想
在筆者講授的量子化學波函數分析與Multiwfn程序培訓班(http://www.keinsci.com/workshop/WFN_content.html)和Multiwfn手冊3.15.5節對HS分析的原理有很具體、詳細的講解,下文只是把原理的最關鍵的部分簡要介紹一下,對于正確做HS分析基本夠了。
HS分析最早由Spackman等人于Chem. Phys. Lett., 267, 215 (1997)提出,后來又得到了發展,Acta Cryst., B60, 627 (2004)和CrystEngComm, 11, 19 (2009)是其兩篇綜述文章。HS分析關鍵思想是對分子晶體中的特定分子構造出Hirshfeld surface,它相當于這個分子在分子環境中的表面,然后再將一些有特殊意義的實空間函數映射到這個表面上,由此可以對分子晶體中的分子間相互作用特征進行考察。
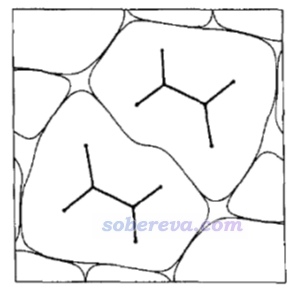
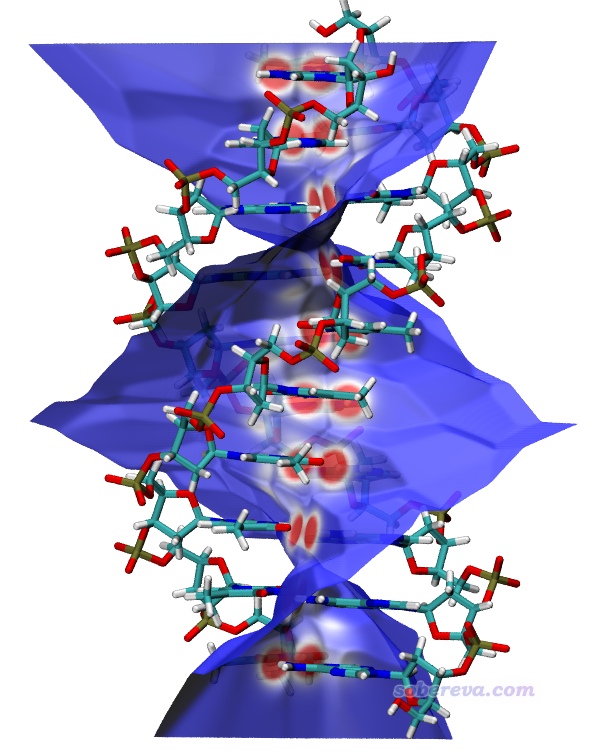
簡要說一下Hirshfeld surface是怎么定義的。Hirshfeld在Theoret. Chim. Acta (Berl.), 44, 129 (1977)最早提出了一種定義化學體系中原子空間的方式,它給每個原子定義了Hirshfeld權重函數來描述這個原子在三維空間中各個位置所占權重,數值從0到1平滑變化,0和1分別對應于此位置完全不屬于和完全屬于這個原子。每個位置所有原子的權重函數加和為1。這是一種典型的模糊式原子空間定義方式。具體定義細節見《原子電荷計算方法的對比》(http://www.whxb.pku.edu.cn/CN/abstract/abstract27818.shtml)的2.5節和Multiwfn手冊3.9.1節,看完了就會知道產生Hirshfeld權重需要有各個原子的坐標,以及體系中的各種元素的原子在孤立狀態下的球對稱化的電子密度。將一個分子中所有原子的Hirshfeld權重函數加和就定義了這個分子的權重,因此分子晶體中各個分子都有各自的權重函數。某個分子的Hirshfeld surface就對應于它的權重函數數值為0.5的等值面。例如下圖的曲線展現了某個平面上各個分子的Hirshfeld surface對應的輪廓。可見,Hirshfeld surface算是分子環境中各個分子與其它分子接觸面的一種定義方式。下文所說的“表面”都是指Hirshfeld surface。
現實當中構造Hirshfeld surface是利用我在J. Mol. Graph. Model., 38, 314 (2012)介紹的Marching Tetrahedron或類似的算法實現的。這個表面被描述為大量小三角形的集合,每個三角形由三個表面頂點構成。由于構造Hirshfeld surface對應的等值面用的算法和《使用Multiwfn的定量分子表面分析功能預測反應位點、分析分子間相互作用》(http://www.shanxitv.org/159)涉及的構造電子密度等值面對應的分子范德華表面的算法在本質上相同,所以Multiwfn中HS分析也是在主功能12(定量分子表面分析)中實現的。
實際中做HS分析的時候是選取一個(也允許是多個)感興趣的分子并對它構造Hirshfeld surface進行分析,這個分子在下文管它叫“中心分子”,它周圍的分子也可以稱為“環境分子”。
HS分析定義了一些映射到Hirshfeld surface上的三維實空間函數,常見的有:
? d_i:表面內部(interior)的原子(也即中心分子的原子)到當前點的最近距離
? d_e:表面外部(exterior)的原子(也即周圍分子的原子)到當前點的最近距離
? d_norm:歸一化的(normalized)分子間接觸距離。由d_i、d_e和原子范德華半徑計算出來。數值越小體現表面上此處附近的內、外原子有越近的接觸,暗示此處的相互作用越強
? shape index(形狀指數):數值在[-1,1]范圍,越負說明此處表面越凹,反之越凸
? curvedness(曲度):在[-4,0.4]范圍,-4對應表面此處完全平坦,越正越凸,0對應單位球面的曲度
d_norm經常用來對Hirshfeld surface進行著色來直觀展現分子環境中的分子間的相互作用,d_norm數值較小的區域對應于較強烈的分子間相互作用。筆者發現用電子密度來著色也很有意義,Hirshfeld surface上電子密度越大的地方體現相互作用越強,效果比用d_norm著色時明顯更好,色彩過渡更為平滑,物理意義也更強,也沒有范德華半徑選取的任意性。由于對大體系做量子化學計算產生比較精確的電子密度比較耗時,因此只需要用準分子近似的電子密度(promolecular density)對Hirshfeld surface著色就夠了,它直接由各個原子孤立狀態的電子密度疊加得到,耗時極低,我實測和使用量子化學計算的電子密度著色的效果差不多。
由于各種元素的孤立狀態的電子密度以及范德華半徑在Multiwfn中是內置的,故產生Hirshfeld surface以及計算以上提及的各種函數只需要原子坐標和元素信息就夠了。由于HS分析依賴的信息非常簡單,不牽扯基于波函數的計算,因此耗時極低,用起來也很方便。
HS分析還經常繪制指紋圖(fingerprint map),是把d_i和d_e作為散點圖的橫軸和縱軸,然后把Hirshfeld surface上的各個頂點根據其d_i和d_e的數值繪制在指紋圖上作為一個個小點。根據指紋圖上散點分布的位置可以對中心和周圍分子間的相互作用特征進行討論,后文會結合具體例子來講。
HS分析中還經常做局部接觸(local contact)分析。完整的HS分析描述了中心分子的所有原子和周圍分子的所有原子間的相互作用,而局部接觸分析可以指定在HS分析中只考慮中心分子的哪些原子和周圍分子的哪些原子的相互作用。比如可以了解在整個Hirshfeld surface中體現中心分子的氧和周圍分子的H之間的相互作用的區域的位置和面積。
Multiwfn還支持Becke surface分析,是我自己提出的概念。它和HS分析的唯一差別是用Becke權重函數而非Hirshfeld權重函數的0.5等值面來定義表面。二者實際效果差別不大,一般沒必要用Becke surface,但它的一個特殊好處是允許表面出現在電子密度為0的區域,此處沒法定義Hirshfeld權重并構造Hirshfeld surface,詳見本文的第5節。Becke權重函數的具體定義方式參見《密度泛函計算中的格點積分方法》(http://www.shanxitv.org/69),它基于原子坐標和原子共價半徑得到。出現在相互作用的原子間的Becke surface會離半徑較小的原子較近、離半徑較大的原子較遠,Hirshfeld surface也有這樣的特點,這是由于定義它的準分子密度分布特征所自然而然帶來的。
2 Multiwfn的HS分析的功能
Multiwfn中做HS分析需要提供含有原子信息的文件作為輸入文件,如.pdb、.xyz、.mwfn、.cif、.fch、.mol2、.gjf等等等等,詳見《詳談Multiwfn支持的輸入文件類型、產生方法以及相互轉換》(http://www.shanxitv.org/379)。
HS分析大多研究的是分子晶體,cif是最常用的記錄晶體結構的格式。通常不能載入cif文件后上來就做HS分析,因為HS分析一般需要提供一個中心分子+環境分子的簇模型,這樣才能靠HS分析考察中心分子與環境分子的相互作用,而cif文件記錄的是晶胞里各個原子的坐標,分子往往是被截斷的,中心分子或環境分子一般都不完整。因此首先需要用《Multiwfn中非常實用的幾何操作和坐標變換功能介紹》(http://www.shanxitv.org/610)中介紹的自動挖團簇的功能構造簇模型。如果你研究的不是分子晶體的情況,就是比如分子二聚體中兩個分子間的相互作用,那么就不牽扯挖簇的過程了,直接提供含有二聚體坐標信息的文件當輸入文件就行了。
HS分析在Multiwfn主功能12實現。進入這個功能后,首先選1把定義表面的方式切換為Hirshfeld surface。此時被映射到表面的函數會自動改為電子密度(對于輸入文件沒有波函數信息的情況具體是指準分子電子密度),你也可以選2把被映射的函數改成其它的。之后選0就開始計算了,Hirshfeld surface會被構造出來,組成它的所有表面頂點上的被映射的函數值會被計算出來,并顯示出Hirshfeld surface的面積和包圍的體積。然后會看到后處理菜單,里面有豐富的選項,提示得都很清楚,Multiwfn手冊3.15.5節都有解釋。利用后處理菜單的選項,可以導出用于在VMD程序中繪制著色的Hirshfeld surface圖要用的.cub文件,還可以繪制指紋圖、做局部接觸分析,這些在后文的例子里都有體現。
后文的例子涉及到的各種文件可以在http://www.shanxitv.org/attach/701/file.zip中獲得。
3 用Multiwfn對NAOB晶體做Hirshfeld surface分析實例
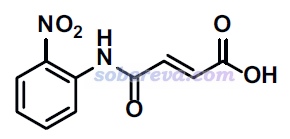
這一節以(Z)-4-((2-nitrophenyl)amino)-4-oxobut-2-enoic acid (NAOB)晶體為例做HS分析。NAOB的分子結構如下,其晶體在DOI: 10.1007/s13738-023-02904-9里進行了研究,文章的補充材料里給了它的cif文件的信息,是本文文件包里的NAOB.cif。
3.0 準備工作:構造簇模型
NAOB.cif對應的晶體結構如下,可見連一個完整的分子都沒有,因此在進行HS分析之前,我們顯然得先構造出中心分子+周圍分子的團簇結構才行。
為了構造簇模型,啟動Multiwfn,然后載入NAOB.cif,之后輸入
300 //主功能300
7 //幾何相關操作
25 //構造“中心分子+臨近一圈分子”的團簇
1 //當前晶胞里1號原子所在的分子將被作為中心分子(由于NAOB晶體里所有分子都是等價的,所以當前隨便輸入一個原子序號即可)
[回車] //代表若一個周圍分子與中心分子間最近原子對距離小于這倆原子的范德華半徑和的1.2倍,則這個周圍分子就被整個納入團簇
Multiwfn瞬間就構造出了團簇,從屏幕上的提示可看到這個簇有375個原子,并且屏幕上還巨貼心地把中心分子中的原子序號給了出來,此例為1-10,17-19,22-25,205-210,214,215。把這個序號記下來,之后HS分析時要用到。
現在可以選當前菜單中的選項0看一眼新構造出的團簇是什么樣,如下所示,可見非常理想,確實是中心分子被周圍一層分子所圍繞(為了中心分子看得清楚,在Multiwfn圖形界面的菜單欄里選了Other settings - Set atom highlighting,然后輸入了前述的中心分子里的原子序號,使中心分子用青色高亮了)。

點圖形界面右上角的RETURN按鈕關閉圖形窗口,然后選擇-2 Output system to .pdb file并輸入NAOB_cluster.pdb,以將當前簇結構導出為當前目錄下的這個文件。這個pdb文件在本文的文件包里也提供了。
注:如《實驗測定分子結構的方法以及將實驗結構用于量子化學計算需要注意的問題》(http://www.shanxitv.org/569)所強調的,由于X光衍射實驗一般難以確定氫原子的準確位置,因此原理上做HS分析之前最好先固定重原子而優化一下所有氫原子的位置。用免費高效的CP2K程序對晶體結構優化氫原子位置然后再用Multiwfn摳團簇,或是先摳團簇再用Gaussian等量子化學程序優化氫,都是可以的。
3.1 繪制電子密度著色的Hirshfeld surface圖
這一節演示繪制基于準分子近似的電子密度著色的Hirshfeld surface圖,這是HS分析最重要的一種圖。除了Multiwfn外還會用到非常流行的VMD可視化程序,可以在http://www.ks.uiuc.edu/Research/vmd/下載,使用筆者現在用的VMD 1.9.3版肯定沒問題,用其它版本不保證能按照本文的例子正常作圖。
啟動Multiwfn,載入上一節產生的NAOB_cluster.pdb,然后輸入
12 //定量分子表面分析
1 //選擇定義表面的方式
5 //Hirshfeld surface
1-10,17-19,22-25,205-210,214,215 //中心分子的原子序號
0 //開始分析
從屏幕上可以看到許多信息,以下兩條是值得注意的,第一個是Hirshfeld surface包圍的體積,相當于分子晶體中屬于這個中心分子的體積,第二個是Hirshfeld surface的面積
Volume: 1644.06256 Bohr^3 ( 243.62494 Angstrom^3)
Overall surface area: 859.01840 Bohr^2 ( 240.54965 Angstrom^2)
現在看到了后處理菜單。選擇-2在當前目錄下導出surf.cub,它是中心分子的Hirshfeld權重的格點數據。再選13,Multiwfn會計算被映射的函數的格點數據并導出為當前目錄下的mapfunc.cub。在這個界面里如果選擇-3也可以直接在Multiwfn里預覽Hirshfeld surface,但沒有著色效果。
將剛剛產生的surf.cub和mapfunc.cub以及Multiwfn自帶的examples\scripts\目錄下的hirsh_rho.vmd作圖腳本一起拷到VMD目錄下(即啟動VMD后在其文本窗口里運行pwd命令看到的目錄)。啟動VMD,在文本窗口里輸入source hirsh_rho.vmd來執行作圖腳本,然后就看到了下圖。
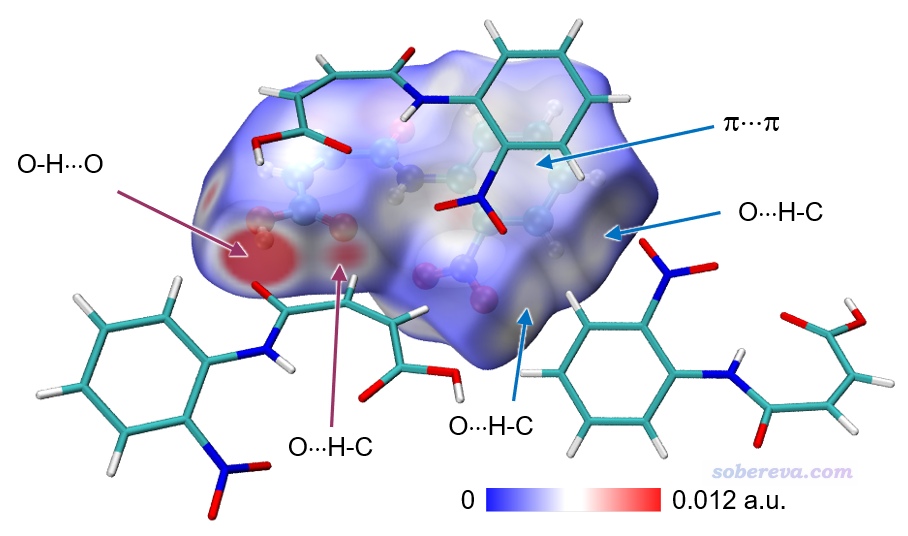
上圖顯示出了中心分子的Hirshfeld surface,并根據電子密度進行了著色。通過hirsh_rho.vmd腳本里的mol scaleminmax top 1 0.0 0.015這條命令可知,默認用的色彩刻度范圍的下限和上限分別為0和0.15。根據腳本里color scale method BWR這行命令可知,當前用的色彩變化是藍-白-紅,色彩變化示意圖在Graphics - Colors - Color Scale里可以看到。因此上圖中越紅的地方就是Hirshfeld surface上電子密度越大的地方,無疑對應于越強的相互作用。上圖中有一塊非常大的紅色,這對應于中心分子羧基的O-H作為氫鍵給體、周圍一個分子的O作為氫鍵受體形成的顯著的氫鍵作用區域。它的旁邊還有一小塊淡紅色區域,對應于中心分子的羧基氧作為氫鍵受體與周圍分子的C-H形成的弱氫鍵。上圖中還有一些發白的區域,在VMD里旋轉圖像仔細觀察的話可以看到對應的是比較遠距離的C-H...O-N很弱氫鍵,以及pi-pi堆積和普通色散吸引作用顯著的區域。這些特征區域都可以自行用powerpoint之類畫個箭頭標注在圖上便于讀者看清楚。上圖還有很多偏藍色的區域,這些地方電子密度接近0,因此不牽扯任何值得一提的分子間相互作用。特別藍的地方也往往對應于分子晶體中的孔洞區域,這種地方電子密度自然特別低,非常建議感興趣的讀者按照《使用Multiwfn計算晶體結構中自由區域的體積、圖形化展現自由區域》(http://www.shanxitv.org/617)介紹的方法作圖考察。
要注意中心分子與各個方向的周圍分子都有相互作用,光是靠一張圖的話很難展現完整,因此文章中可以多給幾張圖展現不同視角的Hirshfeld surface圖。
下面再說一下怎么改進作圖效果。Hirshfeld surface圖的效果受到多方面影響:
(1)光源。可以通過VMD的Display菜單里的Lighting選項設置打開哪些光源。如果選Mouse - Move light,然后在圖形窗口中拖動,還可以移動特點光源的位置。
(2)材質。hirsh_rho.vmd默認對等值面用Translucent材質,可以自行在Graphics - Materials界面里對這個材質的具體定義進行調節。
(3)Graphics - Representation界面里的作圖設置。里面可以創建更多Rep,每個Rep都可以獨立設置顏色和材質,并且通過選擇語句可以定義各個rep顯示的原子,不懂選擇語句怎么寫的話參考《VMD里原子選擇語句的語法和例子》(http://www.shanxitv.org/504)。特別值得一提的是,當前的圖中每個分子都有一個獨立的residue編號并被分子中所有原子所共享,因此可以利用residue選擇特定分子。若想查詢某個分子的residue號,就選Mouse - Query,然后點擊這個分子上任意一個原子的正中央,在VMD的文本窗口中就能看到它的residue號了。
為了讓上面例子的圖像效果更好,我在VMD的Graphics - Representation里做了些修改:把第1個rep設為了residue 0專門用于顯示中心分子,用CPK方式顯示并把Bond Radius設為了0.5。點擊Create Rep按鈕新增一個Rep,選擇語句用residue 7 11 5使得三個與中心分子作用顯著的分子顯示出來,Material設Edgy,Drawing Method用Licorice,把Bond Radius設成0.1。點擊用來顯示Isosurface的那個Rep,再選擇Trajectory標簽頁,在里面把色彩刻度上限設為0.012(注意文本框只顯示兩位小數,輸入0.012后按回車會如實設為0.012),使得著色的色彩顯得更鮮明。在Display菜單里把所有四個光源都打開。最后選File - Render,選擇Tachyon(Internal)進行渲染。之后在圖像上展現特征作用的地方通過powerpoint進行標注,并且把Graphics - Color - Color Scale里顯示的色彩刻度條挪到圖上并適當拉伸、標記上色彩刻度上下限。最終得到下圖,可見對相互作用展現得非常直觀清楚。
如果要繪制d_norm著色的Hirshfeld surface圖,與上面的例子只有兩個差別
(1)進入主功能12并選擇用Hirshfeld surface方式定義分子表面后,選擇2 Select mapped function,再選d_norm。然后再開始分析
(2)作圖使用Multiwfn目錄下examples\scripts\里的hirsh_dnorm.vmd代替hirsh_rho.vmd
由于d_norm著色的圖明顯不如電子密度著色的圖色彩變化平滑,在很多地方有顏色的突越不好看,故這里就不多說了。
Multiwfn還可以給出Hirshfeld surface上被映射的函數(當前為電子密度)的極大點的位置和數值,便于定量對比討論。在Multiwfn后處理菜單中選8 Export all surface vertices and surface extrema as vtx.pqr and extrema.pqr,此時當前目錄下就出現了vtx.pqr和extrema.pqr,分別記錄了所有表面頂點和表面極值點的坐標和被映射的函數數值。如屏幕上的提示所示,extrema.pqr里碳和氧原子分別用于記錄表面極大點和極小點,此文件在本文的文件包里也提供了。按前文用hirsh_rho.vmd作圖后,將extrema.pqr載入VMD,將其顯示方式設為VDW并把Sphere Scale設為0.2,顏色用黃色,此時看到下圖,每個黃色小球都對應Hirshfeld surface上電子密度極大點位置。
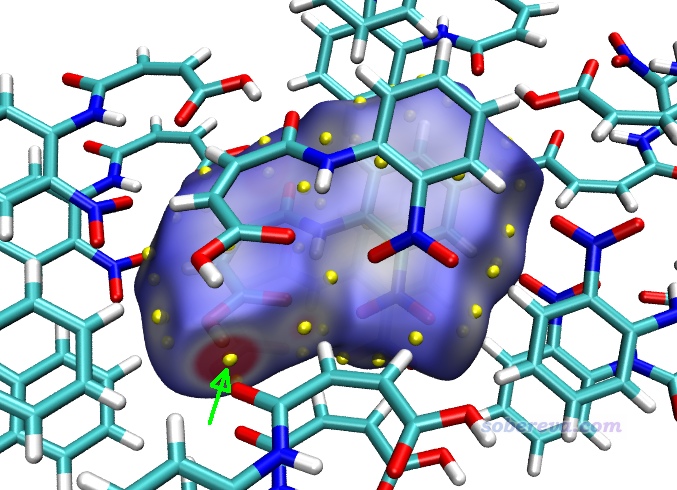
若想查詢表面極值點處的電子密度數值,就在VMD里選Mouse - Query,然后點擊要考察的黃球的正中心,文本窗口就出現了它的index號,從0開始記。上圖箭頭所指的那個對應O-H...O作用的極值點的index為9,相當于從1開始記的編號為10。打開extrema.pqr,找到對應10號碳的下面這一行,倒數第3列的數值0.04228468就是此處的電子密度了,單位為a.u.。
HETATM 10 C MOL A 1 -1.214 3.606 5.014 0.04228468 1.0000 C
以類似的方式查詢旁邊那個C-H...O氫鍵對應的Hirshfeld surface上的電子密度極大點,數值為0.01093053,可見作用顯著弱于O-H...O。
3.2 繪制指紋圖
這一節繪制HS分析中很常見的指紋圖。先按上一節的過程做HS分析并進入到主功能12的后處理菜單,然后輸入
20 //指紋圖與局部接觸分析
0 //開始分析
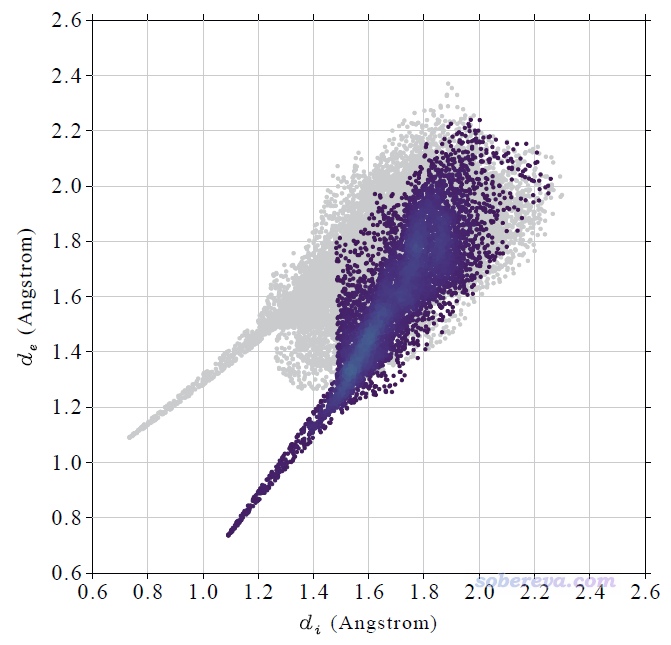
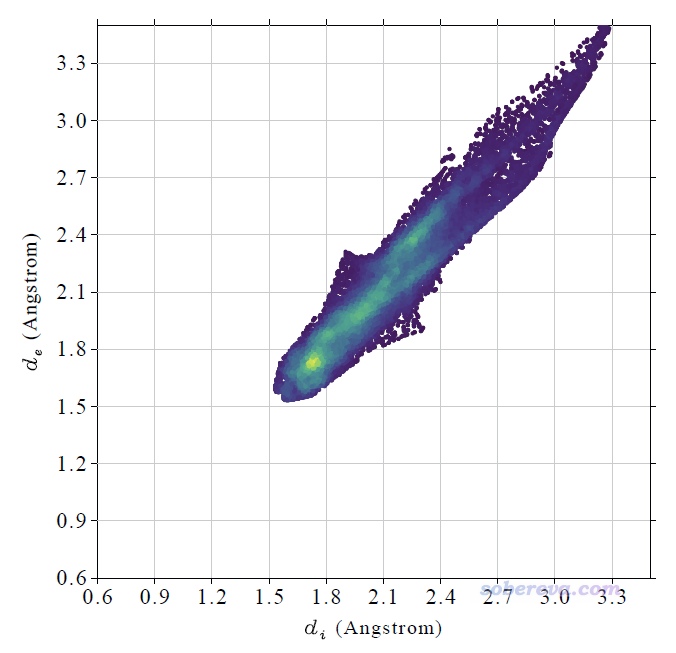
計算很快就完成了,然后看到一個新的后處理菜單,里面的選項不言自明。直接選0就會在屏幕上顯示完整的指紋圖,選1就會把指紋圖保存為當前目錄下的pdf文件(程序用pdf格式是因為它可以無損縮放、線條平滑),此pdf文件已經提供在了本文的文件包里(NAOB_HS.pdf)。當前的圖像如下
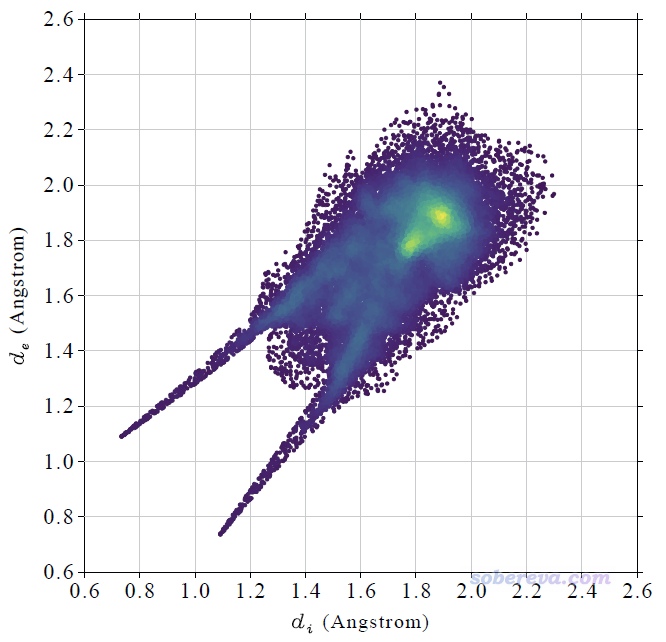
這個指紋圖里每一個小點都是一個Hirshfeld surface上的頂點,并且根據點在指紋圖上的分布密度進行了著色,越密的地方顏色越黃,越稀疏的地方顏色越紫。Multiwfn自動根據指紋圖上的最大分布密度設置色彩變化范圍的上限。Multiwfn畫指紋圖用的這種色彩過度方式稱為viridis,用Google搜圖功能一搜viridis就能找到相應的色條。
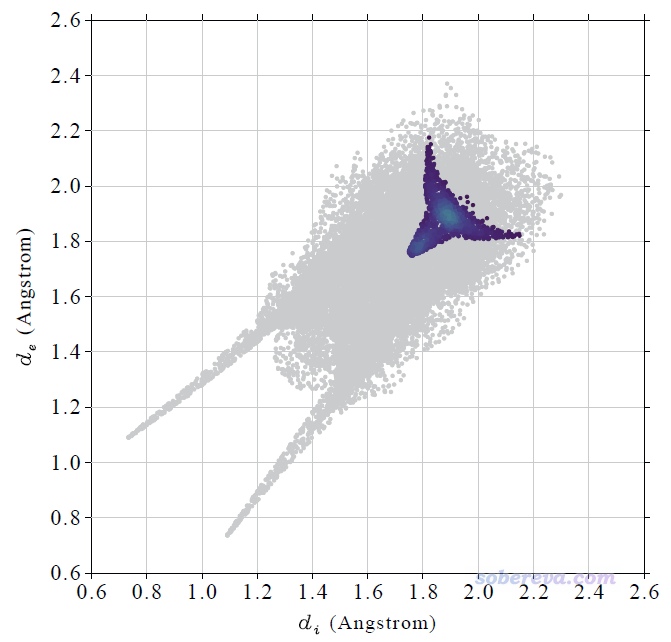
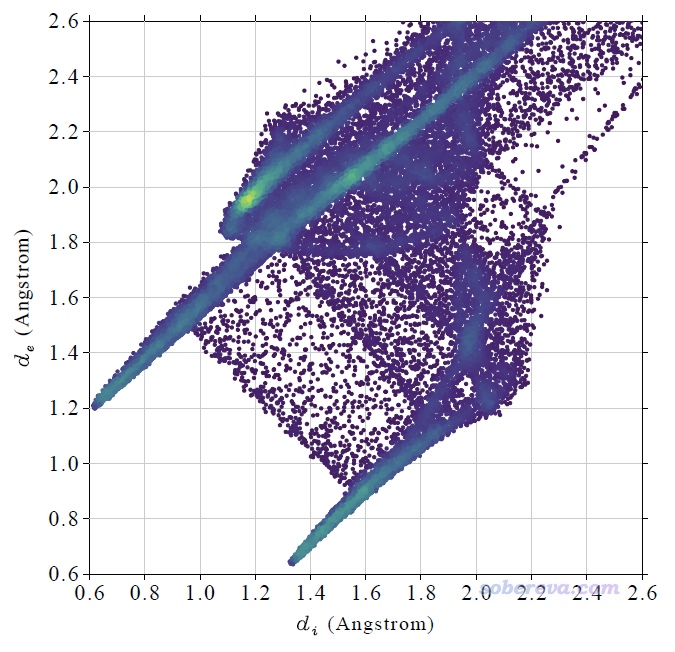
上圖在左下角有兩個顯著的尖兒(spike。兒化音明確體現這是個名詞),是圖像的特征區域,相當于“指紋”。靠左的那個尖兒的頂端大約是d_i≈0.7、d_e≈1.1位置,此處d_e顯著大于d_i,這是此體系具有氫鍵給體特征的體現(如上一節所示此體系的羧基氫確實是氫鍵給體)。為什么這個尖兒的存在能體現此分子存在氫鍵給體?因為這說明在Hirshfeld surface的這個區域,中心分子的原子到表面的最近距離(d_i)顯著小于周圍分子的原子到表面的最近距離(d_e),只有當氫鍵跨越這個區域,半徑很小的氫在表面內、半徑較大的重原子在表面外的時候才會出現這種情況,如下圖所示。
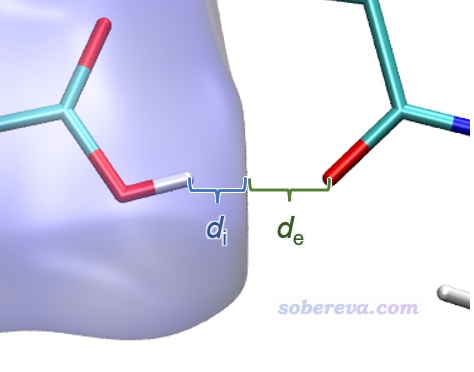
顯然不難理解前面的指紋圖中靠下的那個在d_e≈0.7、d_i≈1.1的尖兒體現的是中心分子作為氫鍵受體與周圍分子形成的氫鍵跨越了Hirshfeld surface。所以指紋圖說明當前研究的NAOB分子同時作為氫鍵給體和氫鍵受體。
指紋圖還蘊含了更多信息,但當前的指紋圖的中間區域密密麻麻一片,很難直接討論,進一步考察就需要利用下一節的局部接觸分析了。
順帶一提,如果你想改變指紋圖上的點的密度,可以在主功能12里做分析之前先選擇3 Spacing of grid points for generating molecular surface修改格點間距,格點間距越小則產生的Hirshfeld surface上的點就越多,指紋圖也會越密。
3.3 局部接觸分析與局部指紋圖
這一節演示一下怎么對中心分子特定的原子與周圍原子特定的原子進行局部接觸分析并繪制與之對應的指紋圖。作為例子,這里考察中心分子的氧與周圍分子的氫的局部接觸情況。
按照上一節說的進入到主功能12的后處理菜單中的20 Fingerprint plot and local contact analyses選項后,輸入以下內容
1 //設置內部原子范圍。默認是所有中心分子的原子都包括
[回車] //代表對原子序號不做限制
O //必須是氧元素
現在中心分子的所有氧原子都納入到了要考慮的內部原子范圍了。然后再輸入
2 //設置外部原子范圍。默認是所有周圍分子的原子都包括
[回車] //代表對原子序號不做限制
H //必須是氫元素
現在周圍分子的所有氫原子都納入到了外部原子要考慮的范圍了。
選擇0開始分析,算完后屏幕上顯示以下信息,告訴你當前考察的這種接觸面積是61.5 Angstrom^2,占Hirshfeld surface總面積的25.57%。
The area of the local contact surface is 61.504 Angstrom^2
The area of the total contact surface is 240.550 Angstrom^2
The local surface occupies 25.57% of the total surface
然后在后處理菜單選擇繪制指紋圖,看到下圖(對應本文文件包里的NAOB_HS_O-H.pdf)。此圖中只有對應于當前考察的局部接觸面上的頂點才被繪制為彩色,可見指紋圖中d_e≈0.7、d_i≈1.1的尖兒確實對應于內O與外H的接觸。
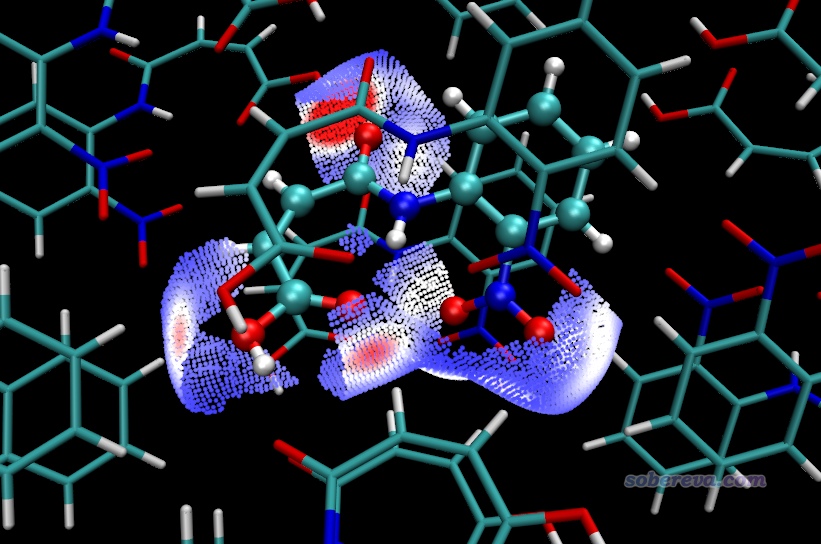
這種局部接觸還可以在立體結構圖上體現。在當前界面里選擇4 Export surface points to .pqr file in current folder,然后當前目錄下就產生了finger.pqr和finger_all.pqr,它們都提供在了本文的文件包里。前者記錄的是當前指定的局部接觸表面上的頂點,后者記錄的是完整的Hirshfeld surface上的頂點。它們可以用文本編輯器打開,可以看到每個表面頂點在pqr文件里用一個碳原子表示,倒數第三列記錄的是Charge屬性,當前被用來記錄表面頂點上被映射的函數數值,對當前來說就是準分子電子密度,單位為a.u.。
為了得到同時展現體系結構和局部接觸表面的圖,現在將NAOB_cluster.pdb載入VMD并恰當設置顯示方式,再把finger.pqr載入VMD,在Graphics - Representation里將它的Drawing Method設為Points并恰當設置Size,Coloring Method設為Charge(即根據Charge屬性著色),在Trajectory標簽頁里把色彩刻度設成與之前繪制等值面圖用的相同的0到0.012。確保Display - Rendermode已經設為了GLSL。在Graphics - Colors - Color Scale里把色彩刻度設為與之前相同的BWR,現在看到的圖如下,確實這些局部表面只對應中心分子的氧和周圍分子的氫的接觸。
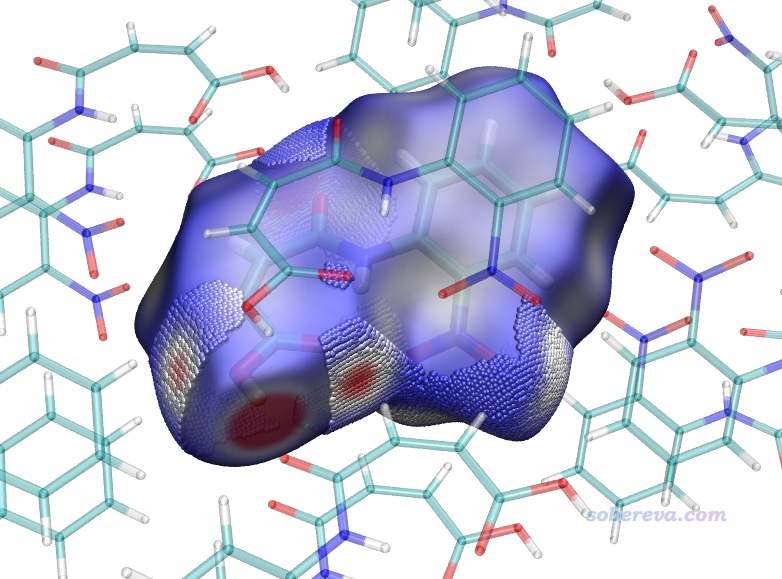
也可以先用hirsh_rho.vmd照常繪制完整的Hirshfeld surface圖,然后再載入finger.pqr,把Drawing Method設為VDW并把Sphere Scale設為0.1,并且對Charge屬性用0到0.012色彩刻度范圍著色,此時看到的圖像如下,局部接觸部分以小球形式著重展現了。
下面再做個演示,對中心分子的芳環和周圍分子的芳環之間考察局部接觸,展現它們之間的pi-pi堆積。在GaussView里,或者在VMD里使用我在《在VMD中顯示原子序號的方法》(http://www.shanxitv.org/197)中提供的腳本,都可以在它們載入NAOB_cluster.pdb后顯示出原子序號,可以看到中心分子的芳環的原子序號是7-9,205,207,209。與中心分子芳環較近因而有pi-pi堆積作用的周圍分子的芳環有兩個,原子序號為11,13,15,112,114,116,201-203,356-358。因此,進入前述的Multiwfn的20 Fingerprint plot and local contact analyses選項后,輸入
1 //設置內部原子范圍
7-9,205,207,209 //原子序號范圍
[回車] //對元素不做限制
2 //設置外部原子范圍
11,13,15,112,114,116,201-203,356-358 //原子序號范圍
[回車] //對元素不做限制
0 //開始分析
4 //導出finger.pqr和finger_all.pqr
然后按照上一節的方法,用hirsh_rho.vmd繪制出Hirshfeld surface,再將finger.pqr載入VMD并恰當設置顯示方式。顯示體系結構的rep的選擇語句寫residue 0 1 11,其中residue 0是中心分子,residue 1和11對應與它有pi-pi堆積作用的上、下兩個分子。然后在Display - Material里把Translucent材質的Opacity改為0.5以降低透明度,并打開Angle-Modulated Transparency選項使得等值面立體感更強一些。之后看到的圖如下,可見小圓球把中心分子芳環上下兩側的pi-pi堆積對應的接觸區域都清晰展示了出來。
在Multiwfn里顯示出相應的局部接觸表面的指紋圖,如下所示。由于碳的原子半徑不小,因此pi-pi堆積對應的局部表面與表面內和表面外的碳原子都有一定距離,而碳原子間距離若太遠也不會有pi-pi堆積效應,因此這些表面頂點在指紋圖中的位置是d_i和d_e都不大不小的區域。
3.4 統計不同元素間的接觸面積
如果想得到中心分子與周圍分子的每一對元素之間的局部接觸面積,雖然通過前面演示的局部接觸分析可以手動實現,但有多少種元素組合就得操作多少次,很麻煩。因此Multiwfn提供了一次性完成的功能。進入前述的Multiwfn的20 Fingerprint plot and local contact analyses選項后,不需要定義內部外部原子,直接選擇選項3 Calculate contact area between different elements,就可以馬上得到以下信息
Inside element, outside element, their contact area (Angstrom^2) and percentage (%)
H-H 47.330 19.676
H-C 10.423 4.333
H-N 0.521 0.216
H-O 56.618 23.537
C-H 12.821 5.330
C-C 22.304 9.272
C-N 2.614 1.087
C-O 4.552 1.892
N-H 0.876 0.364
N-C 2.546 1.059
N-N 0.464 0.193
N-O 1.910 0.794
O-H 61.504 25.568
O-C 4.280 1.779
O-N 1.816 0.755
O-O 9.970 4.145
The same as above, but do not distinguish inside and outside elements
H-H 47.330 19.676
H-C/C-H 23.244 9.663
H-N/N-H 1.396 0.581
H-O/O-H 118.121 49.105
C-C 22.304 9.272
C-N/N-C 5.160 2.145
C-O/O-C 8.831 3.671
N-N 0.464 0.193
N-O/O-N 3.727 1.549
O-O 9.970 4.145
Area of total contact surface is 240.550 Angstrom^2
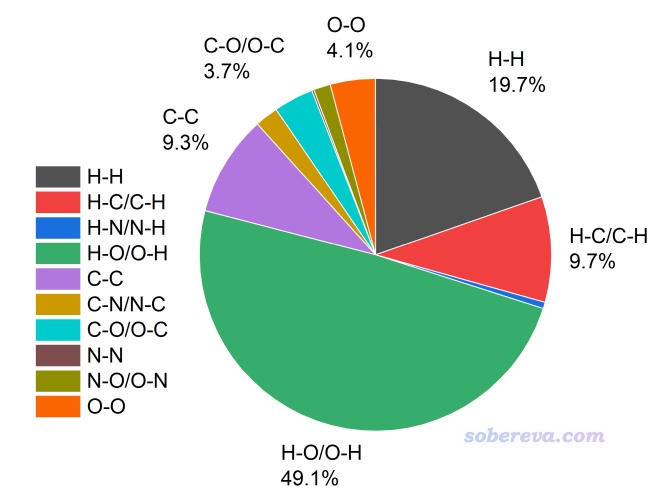
可見以上信息包含了每一對元素的分析結果。例如O-H 61.504 25.568這一行代表中心分子的氧元素與周圍分子的氫元素之間的局部接觸面積是61.504 Angstrom^2,占總面積的25.568%,這和3.4節我們專門算的結果完全一致。而后面輸出的比如H-O/O-H 118.121 49.105代表不管O和H誰在表面內、誰在表面外,兩種元素之間的接觸面積總共是118.121 Angstrom^2,占總面積的49.105%。
為了看起來更直觀,可以把上面的三列形式的數據拷到txt文件里,然后拖到Origin中導入,再繪制餅形圖,如下所示,一目了然。圖中只有占比相對較大的幾種接觸直接標注了數值。
3.5 Hirshfeld surface等值面的截斷問題
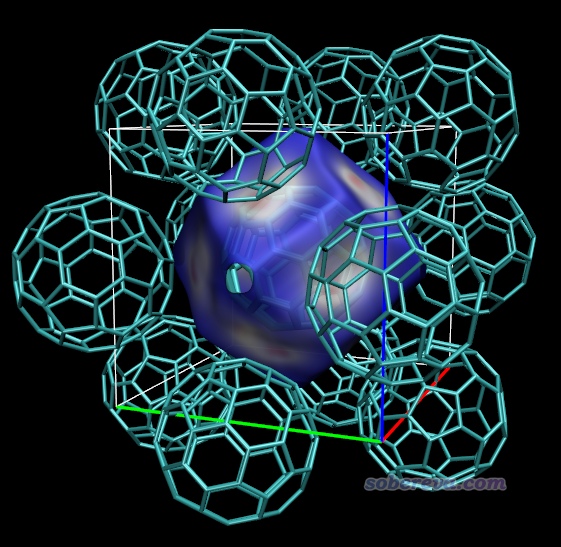
Multiwfn在構造Hirshfeld surface之前,需要先對一個矩形區域(稱為“盒子”)中均勻分布的格點計算你設定的體系片段的Hirshfeld權重。盒子范圍是對你定義的片段往各個方向延展一定距離來確定的,延展距離是位于最邊界的原子的范德華半徑乘以一個系數得到的。默認的系數值是1.7,一般來說夠大了,但如果Hirshfeld surface延伸到距離當前片段較遠的地方,導致超過了盒子范圍,則Hirshfeld surface就會在相應地方被截斷,等值面在那個地方看起來就會有窟窿。此時如果你想要讓等值面完整、完全封閉,顯然就需要增大系數。這一節拿富勒烯晶體舉個例子。
本文文件包里的C60.cif是C60富勒烯晶體結構文件。按照3.0和3.1節的做法摳團簇、繪制電子密度著色的Hirshfeld surface,會得到下圖。這里我在Graphics - Representation里選擇顯示等值面的那個Rep后在界面右下角Show旁邊的下拉框里選擇了Box+Isosurface,這樣除了等值面外,盒子范圍還會用細線同時顯示出來。由下圖可見,等值面上有個難看的窟窿,因為6個富勒烯之間有孔洞區域,Hirshfeld surface實際上會延伸到這里,但當前被尺寸有限的盒子截斷了。
當你發現存在這樣的窟窿,想通過增大延展距離來避免,就應當從主功能12的后處理菜單返回之前的菜單(即剛進入主功能12看到的菜單),然后輸入
4 //高級選項
1 //設置確定延展距離用的范德華半徑的倍數
2.3 //設一個比原本更大的值,數值可以反復嘗試。設得越大,盒子就越大,格點數就越多,計算耗時就越高、產生的cub文件尺寸就越大。由于之前的等值面只有一個小窟窿,所以盒子再大一點就夠了,因此當前嘗試的2.3比之前默認的1.7沒大太多
0 //返回
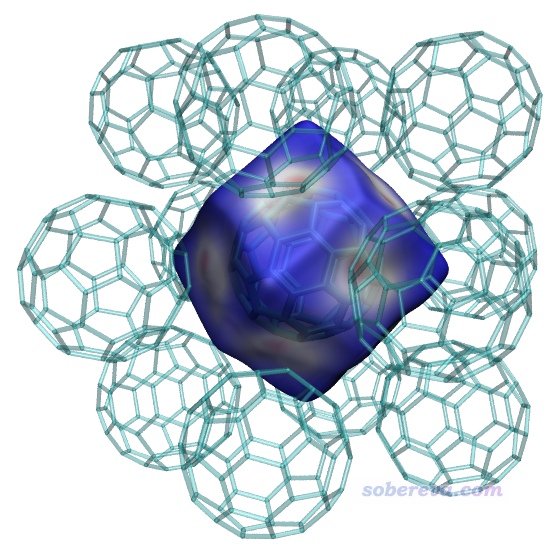
0 //開始計算
之后按照之前的步驟繪制Hirshfeld surface圖,如下所示,窟窿已經沒了。當前的Hirshfeld surface是個正十二面體,每個面都有塊白色且中間偏紅的區域,清楚地展現出中間的富勒烯和周圍富勒烯在此處有顯著的pi-pi堆積作用,由于這些區域離碳原子不遠因此電子密度不是很小。而表面上藍色部分都是晶體中的縫隙、孔洞區域,電子密度極低。
順帶一提,此體系的指紋圖如下所示。繪圖前用Multiwfn菜單里的選項3 Set range of axes適當加大了上限。可見散點的分布區域很窄,各處d_i和d_e數值都差不多大,涵蓋從中等到較大范圍。這體現出表面頂點基本都是位于中間分子和周圍分子之間的正中央區域,并且中間分子和周圍分子有的地方挨得近(d_i和d_e為中等大小),有的地方離得遠(d_i和d_e較大)
4 將Hirshfeld surface分析用于分子復合物
這一節示例HS分析用于展現孤立體系中的片段間相互作用。片段是指一批原子的集合,可以根據需要自由定義,比如既可以是分子復合物中的一個或多個分子,也可以是一個分子中的一個基團,也可以是一個過渡金屬配合物中的一個或多個配體,等等。對于孤立體系,由于感興趣的片段通常不是像晶體環境一樣被其它原子所完整包圍的,所以相應的Hirshfeld surface通常是開放的而不是完全封閉的。
筆者之前做了大量的和18碳環及其衍生物有關的研究工作,匯總見http://www.shanxitv.org/carbon_ring.html。其中研究了具有雙環特征的OPP分子與18碳環的結合問題,成果介紹見《8字形雙環分子對18碳環的獨特吸附行為的量子化學、波函數分析與分子動力學研究》(http://www.shanxitv.org/674)和《理論設計新穎的基于18碳環構成的雙馬達超分子體系》(http://www.shanxitv.org/684)。研究中使用了IGMH方法直觀展現了OPP與18碳環之間的弱相互作用,此例通過繪制電子密度著色的Hirshfeld surface圖也來展現一下。
上述研究中使用DFT優化出來的OPP結合一個18碳環的結構文件是本文文件包里的C18-OPP.pdb。將之載入Multiwfn,然后進入主功能0,會發現此結構文件中18碳環是斜著的,而由于HS分析用的盒子的邊框總是平行于笛卡爾軸的,因此直接對它產生Hirshfeld surface的話表面開口的地方也將是斜著的,看著很惡心。因此此例做HS分析之前還需要先用《Multiwfn中非常實用的幾何操作和坐標變換功能介紹》(http://www.shanxitv.org/610)里介紹的功能令18碳環平行于XY平面,如下所示。
啟動Multiwfn,載入C18-OPP.pdb,然后輸入
300 //其它功能(Part 3)
7 //對當前體系做幾何操作
11 //令一批原子擬合的平面平行于某個笛卡爾平面
225-242 //18碳環的原子序號
1 //平行于XY平面
現在可以選0在圖形界面觀看當前結構,會發現確實18碳環已經在XY平面上了。點RETURN關閉圖形窗口,然后接著輸入
-10 //返回
0 //返回到主菜單
12 //定量分子表面分析
1 //設置表面的定義
5 //Hirshfeld surface
225-242 //18碳環的原子序號
4 //高級選項
1 //設置定義盒子延展距離用的范德華半徑的倍數
2.3 //比默認值更大,實測不這樣的話會導致不好看的截斷
0 //返回
0 //開始分析
-2 //導出surf.cub
13 //導出mapfunc.cub
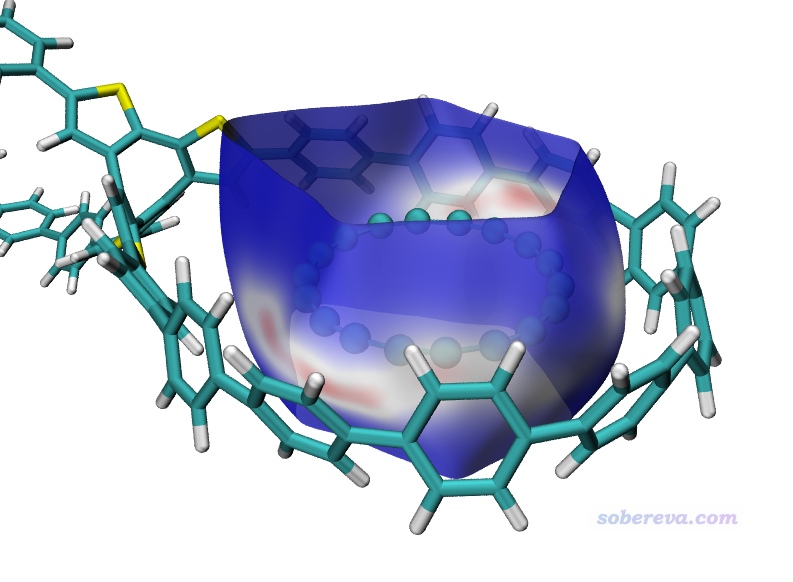
現在基于surf.cub和mapfunc.cub用hirsh_rho.vmd照常繪制Hirshfeld surface圖。這里我把VMD的四個光源都打開了,18碳環用CPK方式顯示,雙環OPP分子用Edgy材質。為了著色效果更鮮明,色彩刻度上限設為了比默認更小的0.01。此時圖像如下所示,可見白色和偏紅的條狀區域把18碳環與OPP的pi-pi作用最強烈的區域展現得挺清楚,此處電子密度比周圍相對更大。由于OPP的大環不是像18碳環一樣理想的圓形,形狀沒有完美匹配,因此它們之間的相互作用明顯不是處處均勻的。通過Hirshfeld surface的顏色可見,在大環末端,特別是兩個大環連接區域,大環與18碳環的pi-pi作用都很弱、電子密度很低。當前的Hirshfeld surface在18碳環上、下方都是開口的,因為在這里被盒子邊界截斷了,顯然這樣的截斷是理應有的,因為上下沒有其它原子了,原理上就不可能是封閉的。
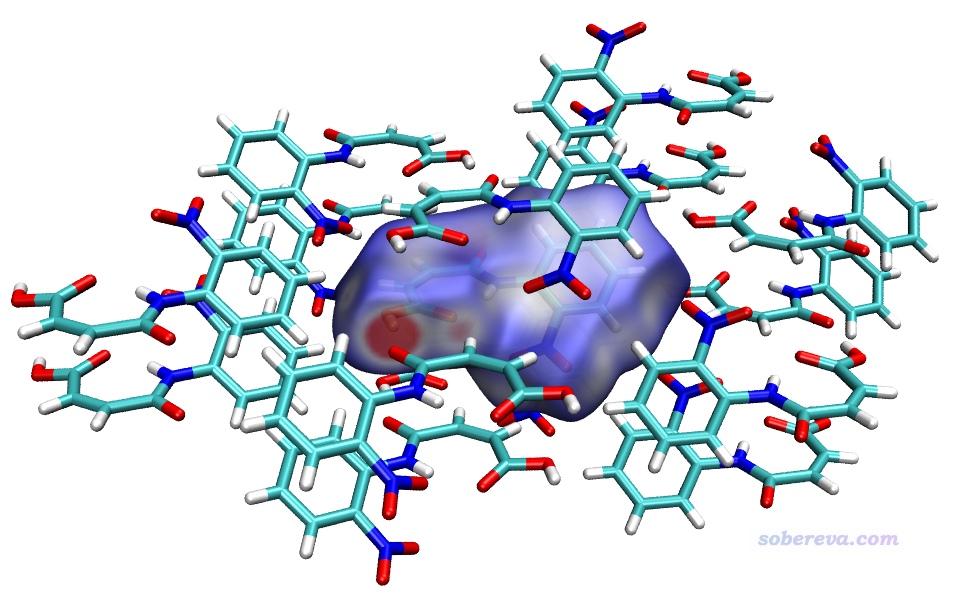
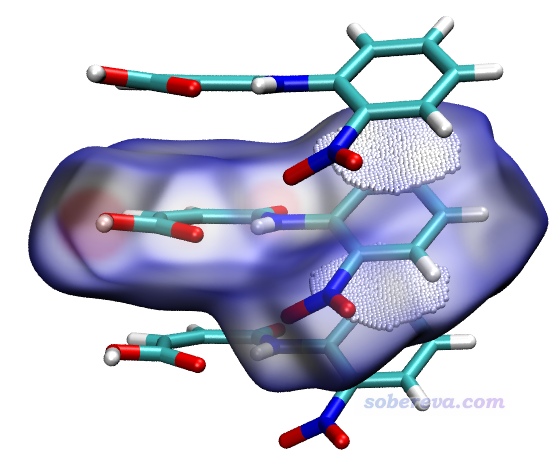
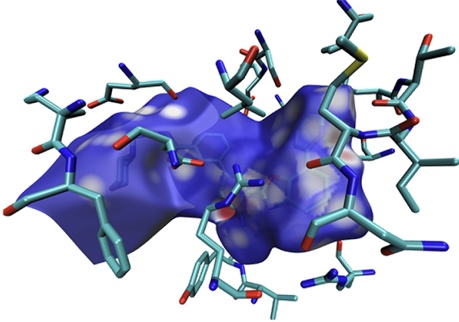
下面這張圖是Comput. Biol. Chem., 101, 107786 (2022)中用Multiwfn+VMD畫的Hirshfeld surface圖,展現了配體和附近的蛋白質的氨基殘基間的相互作用情況,效果不錯,作用位點能看得比較清楚。
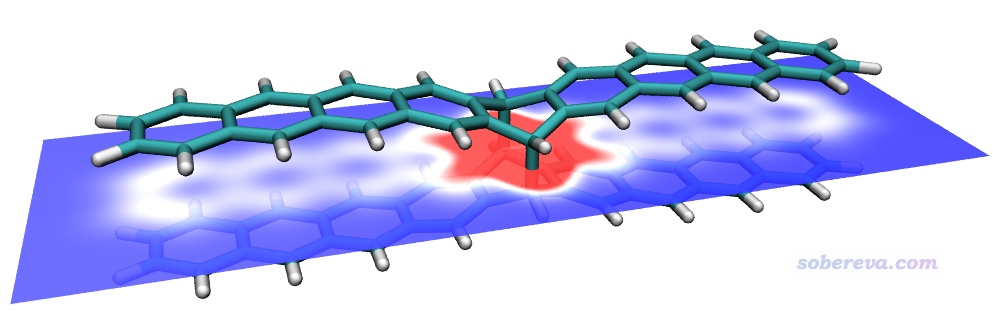
值得一提的是HS分析不僅限于用于考察弱相互作用,讓Hirshfeld surface跨越化學鍵也完全可以。在《一篇最全面介紹各種弱相互作用可視化分析方法的文章已發表!》(http://www.shanxitv.org/667)介紹的我的綜述文章中給出了一個九并苯共價二聚體的例子,這里我也繪制了其電子密度著色的Hirshfeld surface,其中一個九并苯被定義為了片段,色彩刻度用0-0.01,圖像如下所示。可見Hirshfeld surface正好嚴格平行于兩個九并苯、精確處于二者之間。在兩個九并苯之間形成共價鍵的區域及附近電子密度很大,顏色都為紅色,而其它白色區域體現了九并苯之間的明顯的pi-pi堆積作用。
5 Becke surface分析一例:DNA
距離原子非常遠的地方電子密度精確為0,這樣的地方是沒法計算Hirshfeld權重的,Hirshfeld surface也沒法在這個區域出現,這是HS分析的一個缺點。如果極個別情況下被研究的片段與其它部分的接觸面就是會牽扯到距離所有原子都很遠的區域,那么這個時候就必須用我提出的Becke surface代替Hirshfeld surface了,因為在電子密度為0的地方也是可以計算片段的Becke權重的。除了這種極個別情況外沒有用Becke surface的必要,圖像效果不會更好,而且對大體系耗時更高。
這一節就以DNA雙螺旋結構為例繪制做Becke surface分析。Multiwfn自帶的examples目錄下的DNA.pdb是一個DNA片段的結構文件,其中一條鏈的原子序號是1-319,它將被定義為片段。
啟動Multiwfn,載入DNA.pdb,然后輸入
12 //定量分子表面分析
1 //選擇表面的定義
6 //Becke surface
1-319 //第一個片段的原子序號
4 //高級選項
1 //設置定義盒子延展距離用的范德華半徑的倍數
0 //倍數設為0,使得盒子緊貼著邊界原子。因為感興趣的相互作用區域是在兩條鏈間,這樣減小盒子尺寸可以避免Becke surface過大、延伸到不感興趣的區域去
0 //返回
0 //開始計算
在筆者的i9-13980HX筆記本上,用16核并行,花了15分鐘。嫌慢的話可以用核很多的服務器。對于預覽目的,也可以把格點間距設大到比如0.5,耗時只有原本的約八分之一,但此時圖像會明顯更為粗糙。
導出mapfunc.cub和surf.cub并照常用hirsh_rho.vmd繪圖,得到的圖像如下所示,可見Becke surface把兩條DNA鏈間的接觸面非常直觀地展現了出來。在每一層的兩個堿基之間都有兩塊紅色區域,體現出兩個典型氫鍵的存在導致相應地方電子密度相對較大。仔細看的話,會發現有的兩塊紅色區域旁邊還有一小塊白色區域,這對應于C-H...O很弱的氫鍵。Becke surface的其它區域都是藍色,說明兩條鏈在其它地方并沒有值得一提的相互作用(注意不能描述為“沒有相互作用”,用詞必須嚴謹。重原子間的色散作用能即便到了10埃也沒完全衰減到0,兩條鏈的骨架之間的色散作用對相互作用能的貢獻不可完全忽略,只不過強度屬于“不值得專門一提”的程度)
按照HS分析的做法也可以顯示Becke surface的指紋圖,如下所示,可見左下方存在兩個尖兒,體現出每條DNA鏈既作為氫鍵給體也作為氫鍵受體和另一條鏈形成了氫鍵,和HS分析能展現的信息基本一致。但尖兒的具體位置不可能十分一致,畢竟Hirshfeld和Becke權重函數的定義方式差異顯著。
6 總結
本文介紹了HS分析的基本思想,并通過大量例子非常詳細介紹了Multiwfn做HS分析的方法和很多要點。可見HS分析用起來靈活方便,可以較好地直觀展現化學體系的特定片段與周圍其它原子之間的相互作用。同樣的目的用Multiwfn做基于電子波函數的IGMH分析也同樣可以達到,而且IGMH原理更為嚴格,通過sign(lambda2)rho函數對等值面著色還能區分相互作用類型,還能給出各個原子的貢獻量。HS分析可以作為IGMH分析的展現形式的補充,并且由于HS分析只依賴于原子坐標而且計算量很低,因此可以快速地用于很大體系,也不需要事先做量子化學計算產生電子波函數。注:只有原子坐標信息時也可以用Multiwfn做IGM分析,耗時也極低,但圖像效果明顯不及IGMH,參見《使用Multiwfn做IGMH分析非常清晰直觀地展現化學體系中的相互作用》(http://www.shanxitv.org/621)。
最后提醒一下,使用Multiwfn做HS分析或Becke surface分析時請按Multiwfn啟動時的提示恰當引用Multiwfn原文,對于給別人代算的目的也需要明確告知對方這一點。
]]>文/Sobereva@北京科音 2024-Feb-10
在《使用Multiwfn做IGMH分析非常清晰直觀地展現化學體系中的相互作用》(http://www.shanxitv.org/621)中介紹的我在J. Comput. Chem., 43, 539 (2022)中提出的可視化分析弱相互作用的IGMH方法目前已經很流行,在《一篇最全面介紹各種弱相互作用可視化分析方法的文章已發表!》(http://www.shanxitv.org/667)介紹的綜述文章里對此方法也有全面透徹的介紹。
我在去年審了一篇Nature Chemistry的文章,看到作者通過對比對應不同類型相互作用的IGMH等值面來對不對稱催化的原因做了很好的解釋,我覺得很值得作為IGMH分析例子說一下,對一些人可能有啟發。現在這篇文章已經正式發表了:Nature Chemistry, 15, 1672 (2023)。
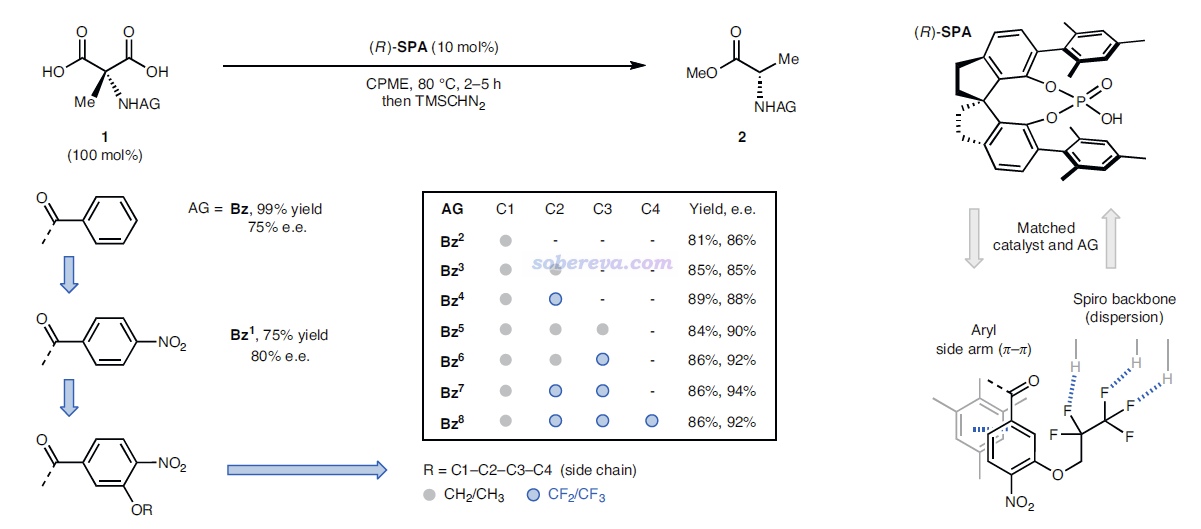
這是一篇實驗為主理論計算為輔的文章,研究對象是下圖的過程。圖的左上角所示的1是個氨基丙二酸類物質,在加入下圖右上角所示的有手性的SPA作為催化劑后,就可以得到脫羧后的結構2,是個有手性的氨基酸類物質。實驗發現,改變底物1中的AG基團會明顯影響產率和對映體過剩率e.e.,如圖所示AG為Bz時是75% e.e.,改為Bz1時80% e.e.,再引入OR取代基后e.e.更高,且R的選擇會進一步影響e.e.,如圖中方框部分所示AG=Bz7時e.e.最高,此時對應于R為CH2-CF2-CF3。下圖右邊示意了這個結構的底物可以與SPA形成顯著的弱相互作用,既有與SPA側鏈的pi-pi堆積作用,底物上的大量的F也與SPA的骨架的C-H有顯著的靜電和色散混合的吸引作用,這些作用可以顯著穩定化SPA對底物的結合。
上面那種AG=Bz7的結構在文中稱為S6,能夠自發脫羧變成下圖的Int-1結構。如下圖左側所示,Int-1可以和S6實現自催化反應(經歷TS-5a),也可以自己發生1-4質子轉移(經歷TS-5b),但勢壘都較高,不是研究的重點。重點是圖中所示的Int-1在SPA的作用下,會經歷一系列過渡態和中間體,最終達到氨基酸類似物結構6',產生S和R手性的這個產物經歷的勢壘有很大差別。圖中可見TS-3是決速步,TS-3S的勢壘遠低于TS-3R,這是SPA催化作用下e.e.很大的關鍵原因。
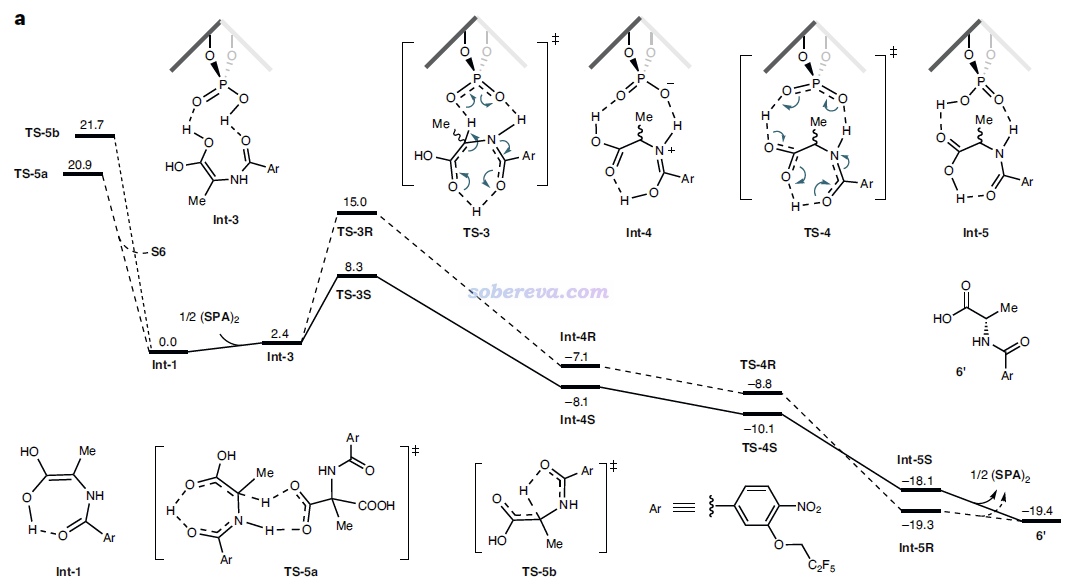
由于反應物相同,因此TS-3S和TS-3R勢壘的差異體現在這兩個結構的能量差別上,差別最主要來自于SPA與Int-1分子的弱相互作用。為了說明這一點,文中用Multiwfn對這兩個過渡態結構做了IGMH分析,給出了下圖,對兩個分子間存在的相互作用類型做了標注。其中N-H...O無疑是挺強的氫鍵,C-H...pi和C-H...O/F都是很弱的氫鍵,苯環間存在pi-pi相互作用,Bz7基團上的一堆F的孤對電子與苯環有色散主導的相互作用。圖中標注Bonding的地方是Int-1與SPA之間發生氫轉移而一定程度成鍵的地方,對TS-3S和TS-3R這個成鍵作用不會有什么明顯差別。
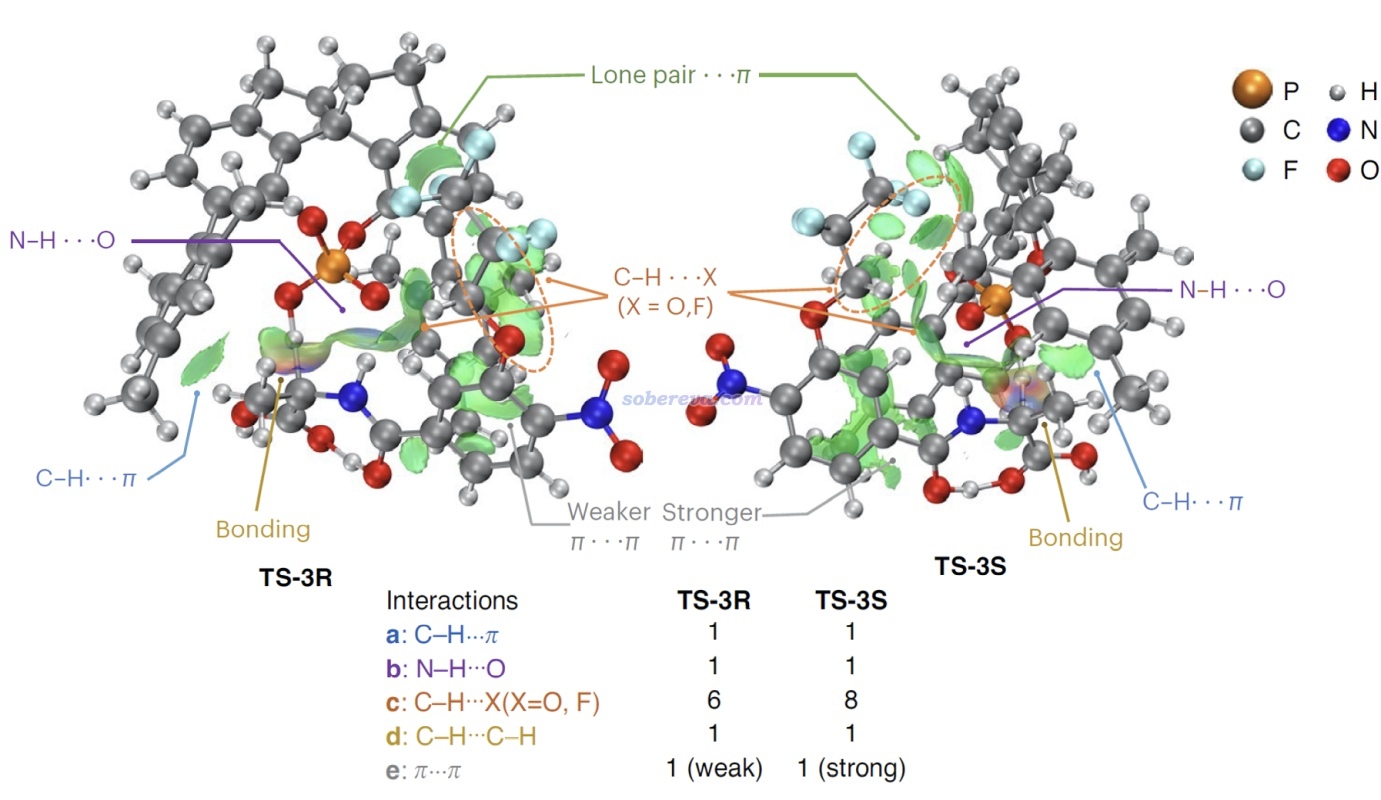
上面兩張IGMH圖的等值面很多,在表面上看起來不太好對比相互作用強度,但如果像本文一樣,把每種相互作用一一羅列出來做細致對比,是完全可以對總作用強度進行區分和解釋的。如上可見,通過仔細觀察等值面可發現TS-3S比TS-3R對應C-H...X作用多出來2個,并且TS-3S的pi-pi堆積作用區域的面積明顯大于TS-3R的,所以前者被標注為strong而后者被標注為weak。
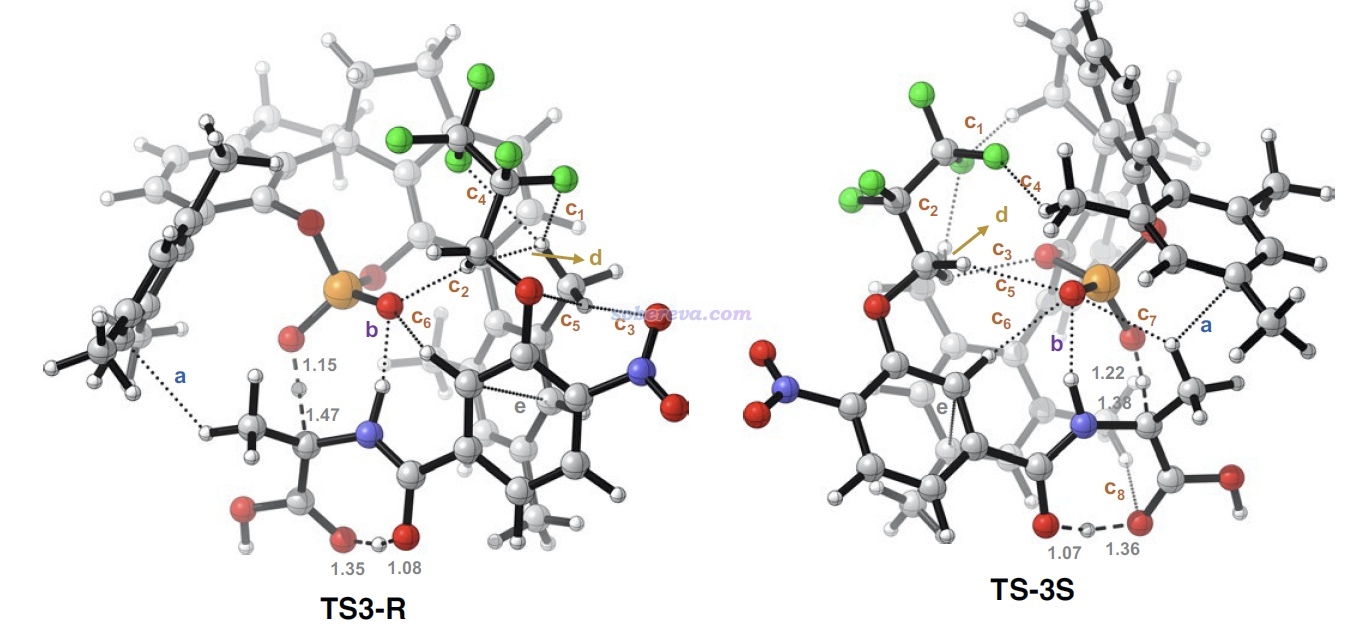
為了能令弱相互作用情況看得更清楚,文中還在分子結構圖上直接用虛線對IGMH呈現的信息做了明確的標注,如下圖所示,字母對應上面的列表里的項,諸如c1、c2...對應不同的C-H...X相互作用。從這個簡化的圖上能更清楚地看到TS3-S中SPA的C-H與底物的Bz7基團上的F之間的相互作用比TS3-R中的更豐富,這一定程度解釋了為什么底物接上F原子較多的Bz7基團時被SPA催化時可以達到較高的e.e.。此外,下面這張圖上還標記了d: C-H...C-H作用的位置,顯然這是普通色散作用,這個作用在上面的IGMH等值面上肉眼不太好辨別(在VMD程序里旋轉等值面時才容易識別),而像下圖這么用箭頭標記一下就容易令讀者看清楚出現在什么位置了。
這篇文章體現出基于IGMH分析能清晰直觀地對分子間相互作用導致勢壘存在的差異和由此帶來的實驗現象予以解釋,是很好的IGMH應用范例,值得在其它研究中借鑒。此文的利用弱相互作用實現e.e.的控制也是新穎的催化設計的思路。
]]>文/Sobereva@北京科音 2024-Jan-26
在《18碳環等電子體B6N6C6獨特的芳香性:揭示碳原子橋聯硼-氮對電子離域的關鍵影響》(http://www.shanxitv.org/696)中提到,筆者在Inorg. Chem., 62, 19986 (2023)一文考察B6C6N6分子的電荷分布時,專門計算了此分子的平面內π軌道、平面外π軌道、σ軌道和內核軌道上的電子是怎么分布在各個原子上的。原子空間定義的方法不唯一,此文用的是流行的Hirshfeld-I原子空間劃分,這種方式劃分的原子空間物理意義較強,可以較合理體現外環境導致的原子空間收縮和膨脹。這種分析方法對于讀者研究很多其它體系也很有益處。本文就演示一下怎么用Multiwfn實現這種計算,以計算B6C6N6的平面內π電子(π-in)的分布為例。由于這個計算需要充分利用Multiwfn的靈活性,牽扯一些細節,這是為什么我專門寫個文章來具體說明。
上述文章考察的B6N6C6的波函數文件是http://www.shanxitv.org/attach/697/B6C6N6_OS.rar,解壓后是B6C6N6_OS.fchk,是由Gaussian 16在wB97XD/def2-TZVP級別下以對稱破缺方式計算得到的。本文例子用的Multiwfn是2024-Jan-21更新的版本。Multiwfn可以在http://www.shanxitv.org/multiwfn免費下載,不了解者看《Multiwfn FAQ》(http://www.shanxitv.org/452)。
啟動Multiwfn,載入B6C6N6_OS.fchk。首先要做的是構造Hirshfeld-I原子空間,把Multiwfn的examples目錄下的atmrad子目錄挪到當前目錄下,這是因為此目錄下有各種元素不同價態的徑向電子密度信息,在構造Hirshfeld-I原子空間時要用到(若缺乏計算機常識不了解什么叫“當前目錄”,看http://www.shanxitv.org/237)。然后在Multiwfn里依次輸入
15 //模糊空間分析
-1 //選擇原子空間
1 //開始構造Hirshfeld-I原子空間
很快就構造完了。Hirshfeld-I原子空間權重數據現已被儲存在了內存里,輸入0返回主菜單。
因為我們要考察π-in分子軌道上的電子分布,故接下來需要把這類軌道以外的分子軌道的占據數都清零。可以按照《使用Multiwfn觀看分子軌道》(http://www.shanxitv.org/269)說的,在Multiwfn主功能0里一個一個觀看占據軌道的圖形判斷,把π-in軌道的序號都記錄下來,最終找到的序號如下。注意當前是對稱破缺計算,因此兩種自旋要分別考察。
Alpha自旋的π-in占據軌道:38,41,42,45,46,48,50,53,54
Beta自旋的π-in占據軌道:596,599,600,603,604,606,608,611,612(注意beta軌道序號在Multiwfn中的記錄規則,在http://www.shanxitv.org/269專門說了)
在Multiwfn里接著輸入
6 //修改波函數
26 //修改軌道占據數
0 //選擇所有軌道
0 //把所有軌道占據數清零
38,41,42,45,46,48,50,53,54,596,599,600,603,604,606,608,611,612 //所有π-in軌道序號
1 //占據數還原為原本的1.0
q //返回
-1 //返回主菜單
現在就可以開始正式計算了。在Multiwfn里接著輸入
15 //模糊空間分析。進入后從選項-1的文字上可以看到當前的原子空間仍是Hirshfeld-I
1 //對特定實空間函數在各個原子空間中積分
1 //被積函數是電子密度。顯然當前對應的是π-in電子密度
馬上看到如下結果
Atomic space Value % of sum % of sum abs
1(B ) 0.64940465 3.607804 3.607804
2(C ) 1.00713759 5.595209 5.595209
3(N ) 1.34345778 7.463655 7.463655
4(B ) 0.64926116 3.607007 3.607007
5(C ) 1.00733834 5.596325 5.596325
6(N ) 1.34338216 7.463235 7.463235
7(B ) 0.64944181 3.608010 3.608010
8(C ) 1.00710182 5.595011 5.595011
9(N ) 1.34348431 7.463802 7.463802
10(B ) 0.64924542 3.606919 3.606919
11(C ) 1.00734233 5.596347 5.596347
12(N ) 1.34339152 7.463287 7.463287
13(B ) 0.64942036 3.607891 3.607891
14(C ) 1.00713369 5.595188 5.595188
15(N ) 1.34344824 7.463602 7.463602
16(B ) 0.64928283 3.607127 3.607127
17(C ) 1.00730639 5.596147 5.596147
18(N ) 1.34341808 7.463434 7.463434
Summing up above values: 17.99999847
Summing up absolute value of above values: 17.99999847
如上可見,B、C、N原子的π-in電子數分別是0.649、1.007、1.343,和如下所示的Inorg. Chem., 62, 19986 (2023)中的表2中的數據完全一致。

最后再提醒一下,必須按以上說明先產生Hirshfeld-I原子空間、修改軌道占據數,最后再在Hirshfeld-I原子空間里積分,而不能先修改軌道占據數然后再進入子功能15產生Hirshfeld-I原子空間并做積分。因為修改軌道占據數之后,此時的電子密度函數就不再是總電子密度了,而Hirshfeld-I原子空間在構造時用到的電子密度函數必須對應總電子密度。
]]>文/Sobereva@北京科音 2024-Jan-26
0 前言
由18個碳原子連接形成的環狀體系18碳環(cyclo[18]carbon)具有十分特殊的幾何和電子結構,自從它于2019年首次在凝聚相中被觀測到后,18碳環及其衍生物就得到了化學界的廣泛關注,并陸續有大量理論研究工作發表。特別是在Carbon, 165, 468-475 (2020)中,北京科音自然科學研究中心(http://www.keinsci.com)的盧天和江蘇科技大學的劉澤玉等人證實18碳環具有明顯的雙pi芳香性特征,這是18碳環非常與眾不同的一點。
18碳環的一種等電子體是B9N9,由B和N原子交替相連構成環狀。此體系早已被研究過,被證實幾乎不具備像18碳環那樣的明顯的芳香性。最近,盧天、劉澤玉和西班牙多諾斯蒂亞國際物理中心的Mesías Orozco-Ic發現,由6個(硼-碳-氮)重復單元依次相連構成的18碳環的另一種等電子體B6C6N6具有和18碳環很接近的芳香性。為什么在無芳香性的B9N9中摻雜進去碳原子后,或者說讓碳原子橋聯硼和氮后,B9N9的芳香性就能得到巨大提升?這是非常有意思的問題。于是以上研究者對B6C6N6的特征做了全面深入的理論研究,詳細分析討論了其芳香性特征和本質,成果近期在美國化學會的知名期刊Inorganic Chemistry上發表,非常推薦閱讀:
Exploring the Aromaticity Differences of Isoelectronic Species of Cyclo[18]carbon (C18), B6C6N6, and B9N9: The Role of Carbon Atoms as Connecting Bridges, Inorg. Chem., 62, 19986 (2023) https://doi.org/10.1021/acs.inorgchem.3c02675
此研究的研究意義不僅在于討論了B6C6N6本身,還在于揭示了碳原子橋聯硼和氮原子對電子離域特征的關鍵性影響,這對于通過碳摻雜的方式對純硼、氮構成的體系(如硼-氮納米管、二維層狀h-BN體系等)進行改性提供了重要啟示。此研究通過強大的Multiwfn程序(http://www.shanxitv.org/multiwfn)利用了各種波函數分析手段對B6C6N6進行了充分的考察并和18碳環、B9N9進行了對比,這也是充分運用波函數分析探究新穎體系的很好的范例。
下面本文就對Inorg. Chem., 62, 19986 (2023)這篇文章的主要研究思想、結論進行深入淺出的介紹,并對一些研究細節進行附加說明,以便于讀者更好地理解此文,以及將此文的研究手段用于自己的研究。同作者之前還對18碳環及其衍生物的各方面特征做過充分的理論研究并得到了同行的廣泛關注,成果匯總見http://www.shanxitv.org/carbon_ring.html。
1 B6N6C6的幾何結構
做B6N6C6的各方面研究之前先得得到其可靠的幾何結構。18碳環及衍生物用ωB97XD/def2-TZVP級別做幾何優化是很穩妥的,在Carbon, 165, 468-475 (2020)以及http://www.shanxitv.org/carbon_ring.html里列舉的此類體系的大量研究工作中都已經充分證明了這點。因此此文用Gaussian 16在此級別下優化了B6N6C6的結構。特別需要注意的一點是,B6N6C6基態是單重態,但是其閉殼層單重態波函數存在不穩定性,用DFT計算時需要作為對稱破缺單重態來處理方可得到真正的基態,相關討論見《談談片段組合波函數與自旋極化單重態》(http://www.shanxitv.org/82)。因此,此文對B6N6C6基態的各種研究都是基于對稱破缺單重態的結構和波函數做的。如果誤當成了閉殼層單重態來算,作者發現得到的幾何結構將明顯不合理,而且電子結構也明顯錯誤,比如芳香性嚴重偏高。實際上之前有一篇論文也算過B6N6C6,但由于那些作者沒注意這一點而誤當成了閉殼層計算,導致結果是沒有任何意義的。此例體現出DFT計算B6N6C6這種電子結構不同尋常的體系一定要注意做波函數穩定性測試。而ωB97XD/def2-TZVP級別下的B9N9、18碳環的穩定波函數都是單重態閉殼層的。
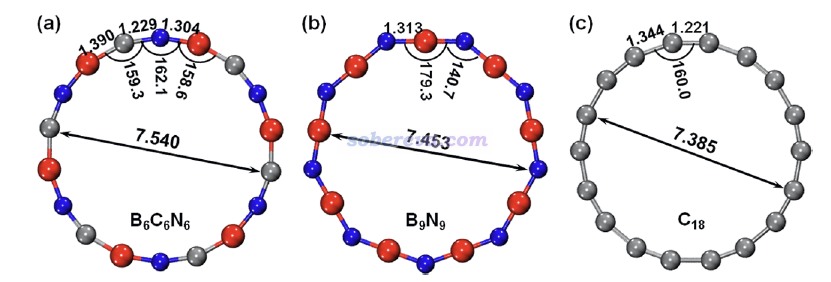
ωB97XD/def2-TZVP級別下優化得到的無虛頻的B6N6C6、B9N9和18碳環的結構如下所示,紅/灰/藍分別對應硼/碳/氮,笛卡爾坐標在文章的SI里提供了。可見B6N6C6屬于C6h點群,而B9N9屬于D9h點群。
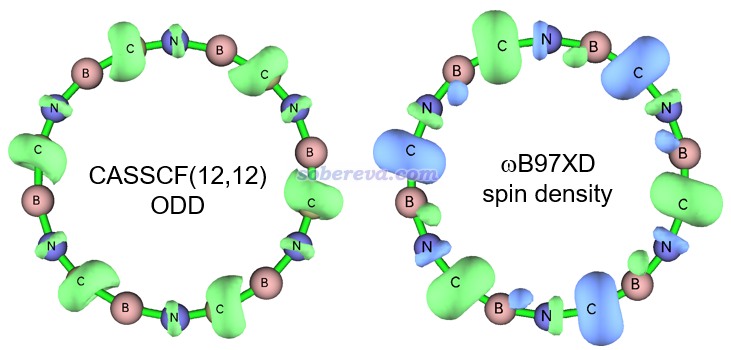
畢竟B6N6C6是一個新穎的結構,不排除靜態相關很顯著導致ωB97XD描述不理想的可能,故為了絕對穩妥,此文還用ORCA在CASSCF(12,12)/def2-TZVP級別下也做了幾何優化。此活性空間設定下,最終會有12個占據數明顯偏離整數的pi型自然軌道,而若活性空間進一步擴大到CAS(14,14),則會再多出兩個占據數接近整數的pi型自然軌道,這體現CAS(12,12)恰好足夠充分描述B6N6C6的pi電子的靜態相關效應。CASSCF(12,12)優化出的結構和ωB97XD的非常相近,體現出ωB97XD得到的此體系的幾何結構靠譜。為了進一步體現ωB97XD計算出的波函數也合理,文中也順帶對比了一下這兩個級別得到的單電子分布情況。文章按照《談談自旋密度、自旋布居以及在Multiwfn中的繪制和計算》(http://www.shanxitv.org/353)計算了ωB97XD波函數的自旋密度,而由于CASSCF沒法得到自旋密度,故文章使用《使用Multiwfn計算odd electron density考察激發態單電子分布》(http://www.shanxitv.org/583)介紹的方法對CASSCF(12,12)波函數產生了單電子密度(ODD),等值面圖對比如下所示。在不考慮自旋密度符號(體現在等值面顏色上,不同顏色對應單電子不同自旋方向)的情況下,可見二者展現出的單電子分布是基本吻合的,即單電子主要都分布在碳上。這在很大程度上體現出ωB97XD對稱破缺計算產生的波函數也是合理的,適合用于之后的波函數分析。
2 B6N6C6的電荷分布
原子電荷是定量考察化學體系中電荷分布最常用的指標之一。18碳環由于所有的碳都是等價的,顯然原子電荷都為0,而對B6N6C6和B9N9計算原子電荷,可以直接考察這倆體系中不同原子帶的凈電量、了解二者電荷分布的差異。原子電荷計算方法并不唯一,不同方法基于不同的思想、有不同的特點,在《原子電荷計算方法的對比》(DOI: 10.3866/PKU.WHXB2012281)中有大量介紹和對比。考慮到B6N6C6電子結構的特殊性,為了得到穩妥的結論,文中用Multiwfn計算了很常用的ADCH、Hirshfeld、Hirshfeld-I、CHELPG原子電荷(它們的計算例子看Multiwfn手冊4.7節)、用NBO程序計算了常見的NPA原子電荷,結果如下所示。可見雖然不同原子電荷給出的具體數值不同,但可以得到共同的結論,就是氮明顯帶負電,硼明顯帶正電,這體現出了它們顯著的電負性差異。而在B6N6C6中,硼和氮的凈電荷差異明顯小于它們在B9N9中,這體現出碳元素在硼、氮之間的插入明顯促進了體系整體電荷分布的均衡性。而B6N6C6中碳自己的原子電荷則接近于0,即沒怎么受周圍化學環境的影響。
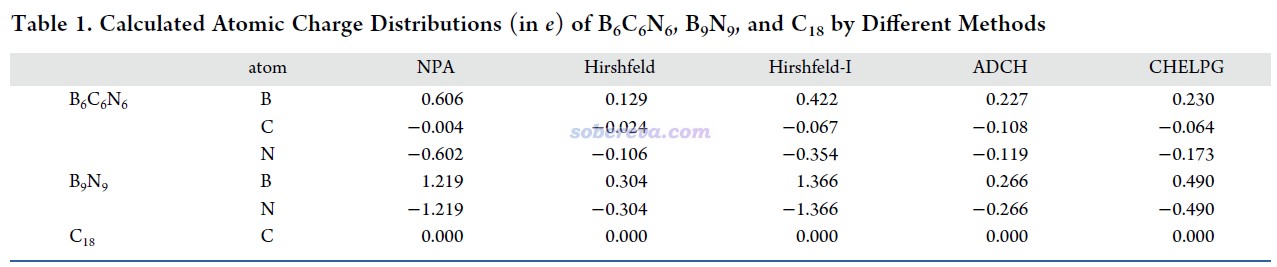
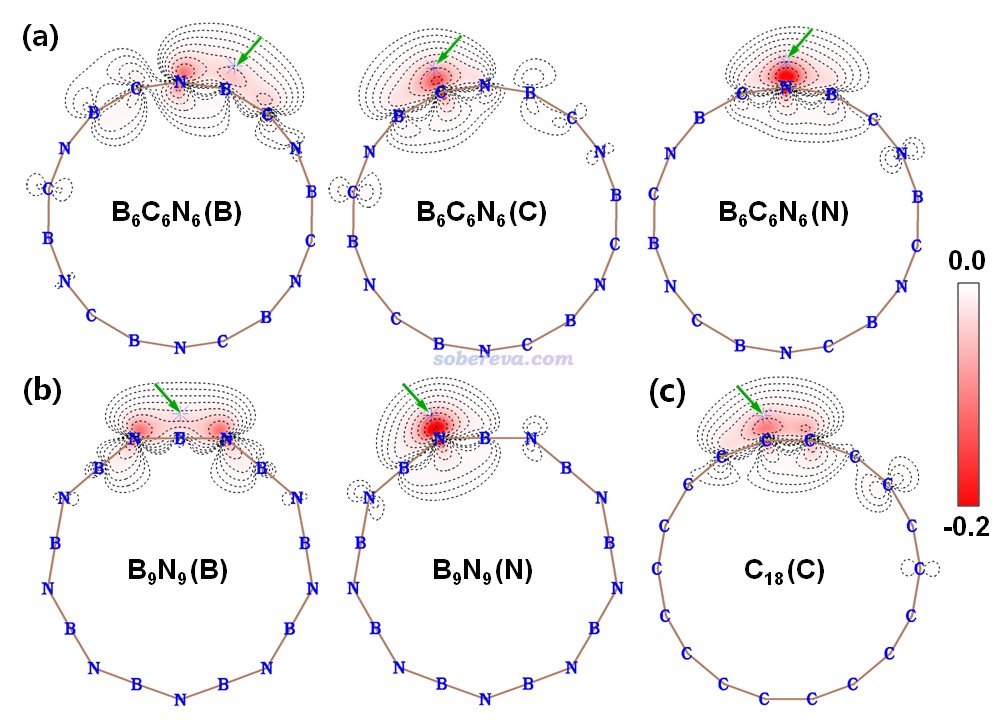
18碳環具有四類分子軌道:平面內和平面外兩套π軌道,分別稱為π-in和π-out,此外還有σ軌道,以及碳的內核原子軌道組成的內核分子軌道。B6C6N6和B9N9也有這四類軌道。為了深入考察各個原子上的電子是怎么分布在這些軌道上的,文中用Multiwfn基于Hirshfeld-I原子空間劃分對三個體系計算了電子在這些軌道上的布居數,如下所示。具體計算方法我專門寫了篇文章說明:《使用Multiwfn基于Hirshfeld-I劃分計算特定類型電子在各個原子上的分布量》(http://www.shanxitv.org/697)。
從以上數據可以看到,在每個原子上,π-in和π-out電子分布量都是基本一致的,沒有什么偏向性,體現出這些體系里π-in和π-out電子特征的近似等價性。B6C6N6和18碳環中的碳的電子結構特征非常相近,π-in和π-out電子數在兩種體系里都基本為1.0,明顯都處于sp雜化的狀態。
3 B6N6C6的成鍵情況
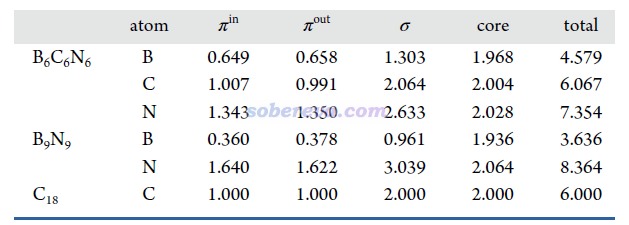
鍵級是考察化學鍵特征非常常見的方式,參見《Multiwfn支持的分析化學鍵的方法一覽》(http://www.shanxitv.org/471)。文中用Multiwfn對B6C6N6、B9N9和18碳環計算了流行的Mayer鍵級,并且分解為了不同類型軌道的貢獻,如下所示。Mayer鍵級體現了原子間等效的共享電子對數,由數據可見,三個體系里所有鍵都包含典型的單個σ鍵特征(即σ+core電子貢獻的Mayer鍵級接近非常接近于1),同時π-in和π-out電子都構成了一定程度的π鍵特征,且二者貢獻相近。B6C6N6中的π作用程度是N-B ≈ C-N > B-C。B9N9中所有B-N等價。18碳環由長、短兩種C-C鍵構成,后者的π作用顯著強于前者。文中還用Multiwfn計算了模糊鍵級以進一步確認了這些結論,數據見補充材料。
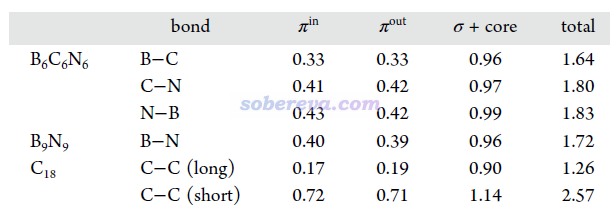
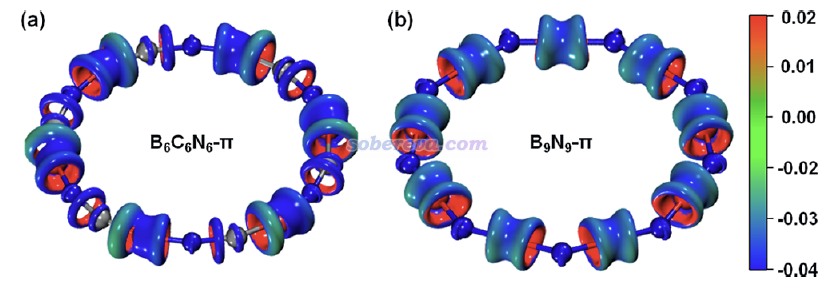
《使用IRI方法圖形化考察化學體系中的化學鍵和弱相互作用》(http://www.shanxitv.org/598)中介紹了盧天提出的IRI-π函數,可以直觀地展現化學體系中π電子相互作用情況。此文對B6C6N6和B9N9繪制了IRI-π=1.2的等值面圖并用sign(λ2)ρ函數著色,如下所示。可見每個鍵上都出現了環狀的等值面,非常直觀地體現出這些鍵都存在明顯的π作用,這和Mayer鍵級展現出的信息相吻合。
4 B6N6C6的芳香性
B6N6C6的芳香性是當前研究的重點。和18碳環一樣,B6N6C6和B9N9的π-in和π-out電子皆為18個,都滿足Hückel的4n+2芳香性規則,因此都有可能存在雙π芳香性。衡量芳香性的方法非常多,在《衡量芳香性的方法以及在Multiwfn中的計算》(http://www.shanxitv.org/176)里有充分的介紹。其中基于電子結構和基于磁性質的芳香性衡量方法的可靠性和普適性普遍較好,可以用于考察B6N6C6的芳香性并與18碳環和B9N9的進行對比。
AV1245*1000在《使用Multiwfn計算AV1245指數研究大環的芳香性》(http://www.shanxitv.org/519)里專門介紹過,是基于波函數定義的適合用于定量考察共軛大環體系芳香性的指數。文中首先使用Multiwfn計算了AV1245*1000,結果如下。除了總值外,還分別給出了π-out電子和其它電子的貢獻(注:π-in、σ、core電子對AV1245的貢獻無法精確分離故沒有單獨給出。但顯然σ和core的離域性極低,不會對AV1245有什么貢獻,故“其它電子貢獻”實質上等于π-in電子的貢獻)。由數據可見,B6C6N6的芳香性顯著強于B9N9,并且π-out的芳香性比π-in還略強一點,這可能在于體系的π-in電子的共軛程度相對略低,源自于環的曲率使得平行于環的p原子軌道彼此交疊程度低于垂直于環平面的p原子軌道。單從AV1245*1000的數值來看,B6C6N6的芳香性甚至比18碳環還要更強一點。

NICS是非常流行和穩健的基于磁屬性衡量芳香性的指標,它的不同形式在http://www.shanxitv.org/176里有專門介紹。其中NICS(0)zz對本文研究的這些環狀共軛體系最為適合,結果如下所示,數值越負說明芳香性越強。為了更充分了解芳香性的來源,文中還按照《基于Gaussian的NMR=CSGT任務得到各個軌道對NICS貢獻的方法》(http://www.shanxitv.org/670)介紹的方法分別計算了π-in、π-out和σ+core電子的貢獻。NICS(0)zz體現出B6C6N6的芳香性接近18碳環,并且π-in和π-out的貢獻相仿佛。而B9N9雖然形式上也滿足4n+2規則,但由于其NICS(0)zz基本為0,因此可斷定是非芳香性的,這必然是由于其π電子缺乏整體離域能力所致。雖然B9N9的各個B-N鍵如前述Mayer鍵級和IRI-π所示有明顯π作用,但顯然不意味著其π電子能夠容易地離域。對這些體系,σ和core電子都由于其高度定域性而對芳香性基本沒有影響。
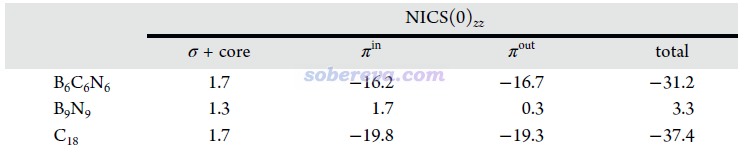
《使用AICD程序研究電子離域性和磁感應電流密度》(http://www.shanxitv.org/147)和《使用AICD 2.0繪制磁感應電流圖》(http://www.shanxitv.org/294)介紹的AICD圖是使用得較普遍的圖形化直觀展現化學體系磁感生電流的方法。為了進一步從磁感生電流的角度展現B6C6N6的芳香性以及和B9N9的差異,文中對這些體系的π-in、π-out以及全部π電子分別繪制了AICD圖,如下所示。外磁場方向垂直于環由下朝上施加。由于等值面上的箭頭太小,為了看得清楚,圖中還用大的橙色箭頭做了清晰的標注。此圖直觀體現出,B6C6N6確實存在顯著的π芳香性,因為感生電流方向符合左手定則并且連貫地環繞整個體系,這正是典型的π芳香性體系具備的特征。而缺乏芳香性的B9N9則明顯不具備這樣的特征,感生電流幾乎只是繞著原子核或原子間區域轉。
此文還按照《考察分子磁感生電流的程序GIMIC 2.0的使用(含24分鐘演示視頻)》(http://www.shanxitv.org/491)介紹的方法繪制了GIMIC磁感生電流的動畫,如下所示,更進一步地體現出芳香性明顯的B6C6N6能夠形成整體感生環電流而非芳香性的B9N9不具備這樣的特征。
B6C6N6:http://www.shanxitv.org/attach/696/B6C6N6_GIMIC.mp4
B9N9:http://www.shanxitv.org/attach/696/B9N9_GIMIC.mp4
文中還對穿越B6C6N6、B9N9和18碳環的化學鍵的感生電流進行了積分,數值分別為21.64、0.70、22.50 nA/T,這定量體現出B6C6N6和18碳環的芳香性相仿佛,而B9N9完全不具備芳香性。
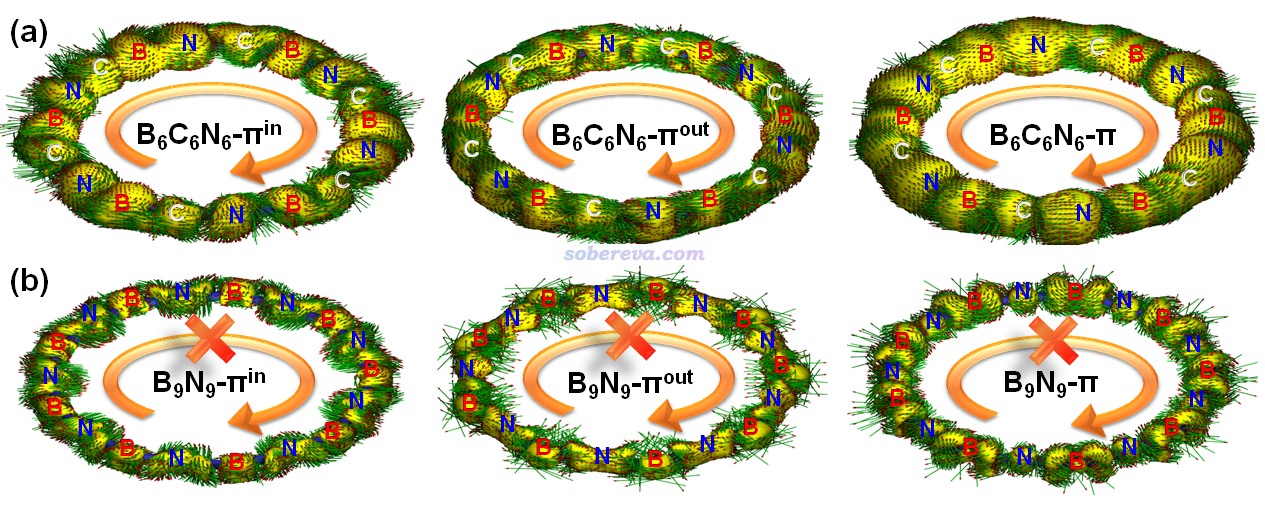
在《通過Multiwfn繪制等化學屏蔽表面(ICSS)研究芳香性》(http://www.shanxitv.org/216)和《使用Multiwfn巨方便地繪制二維NICS平面圖考察芳香性》(http://www.shanxitv.org/682)中介紹了ICSS函數,它直觀地體現了化學體系的磁屏蔽和去屏蔽區域的位置和強度,是NICS的重要擴展,已被非常廣泛應用于討論芳香性問題。此研究使用Multiwfn對B6C6N6和B9N9都繪制了ICSS_ZZ = 10 ppm的等值面圖以及垂直于環的截面的填色等值線圖,如下所示。等值面圖中橙色和藍色分別對應屏蔽和去屏蔽區域。由圖可見,B6C6N6的環內部完全是屏蔽區域,環外面完全是去屏蔽區域,這種特征和苯這樣的典型芳香性體系完全相同,更進一步嚴格地證明了B6C6N6有顯著的芳香性。而且B6C6N6的ICSS的這種特征和Carbon, 165, 468-475 (2020)報道的18碳環的高度一致,體現出二者芳香性的高度共性。而B9N9的ICSS等值面圖的屏蔽和去屏蔽區域彼此交錯,沒有形成遍及整體的連貫的等值面,體現出此體系既沒有芳香性也沒有反芳香性,而是非芳香性。
5 B6N6C6的電子離域性
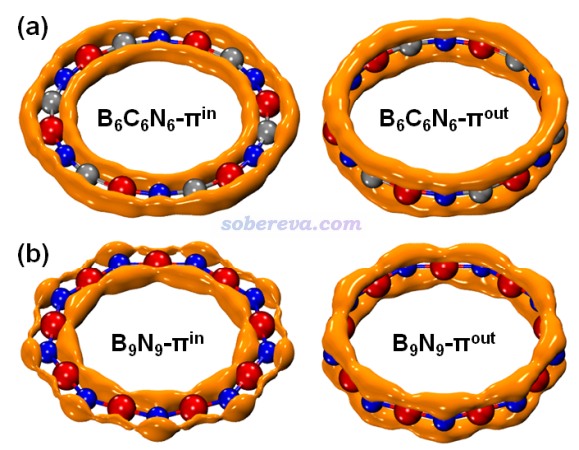
芳香性的本質來自于電子的離域,因此文中還特意對B6N6C6的電子離域性從多方面進行了考察。在《在Multiwfn中單獨考察pi電子結構特征》(http://www.shanxitv.org/432)中介紹了Multiwfn可以計算和繪制的LOL-π函數,這對于考察π電子的離域情況極其有用,如今已經非常流行。下圖對比了B6N6C6和B9N9的LOL-π=0.5等值面,明顯可見B6N6C6的等值面比B9N9顯得連貫得多,明顯體現出B6N6C6的π電子有強得多的在18個原子上全局離域的能力,也一定程度解釋了前者的芳香性比后者強得多的原因。
LOL-π函數比較適合直觀定性考察,而《在Multiwfn中單獨考察pi電子結構特征》里介紹的ELF-π函數的二分點數值則適合定量分析對比。下圖是B6N6C6和B9N9的ELF-π函數等值面圖,等值數值設為了令等值面剛好分裂時的數值,這被稱為二分值,數值越大說明電子越容易跨越二分點區域發生離域。在圖上也標注了二分點位置和二分值。由圖可見,B6N6C6的π-in和π-out的二分值都明顯大于B9N9的,這給B6N6C6的整體電子離域性更強又進一步提供了定量證據。

電子的費米穴是考察電子離域特征的一個重要的函數,它包含兩個三維位置坐標,作圖考察時通常將一個位置作為參考點,然后對另一個坐標繪圖。Atoms-in-molecules理論的提出者Bader指出an electron can go where its hole goes and, if the Fermi hole is localized, then so is the electron,也就是假設一個電子出現在某個參考點位置,那么其費米穴函數明顯分布在哪,這個電子就容易離域到哪去。因此,通過繪制費米穴圖,可以更進一步了解體系處于不同位置的電子的離域能力。文中通過Multiwfn對B6N6C6、B9N9和18碳環分別繪制了費米穴的二維圖,如下所示。參考點位置以綠色箭頭標注,設在了不同原子的價層區域距離原子核特定距離的位置,此時圖中的費米穴的分布就體現了這個位置的價電子容易離域到哪去。用Multiwfn繪制這種圖的操作在《制作動畫分析電子結構特征》(http://www.shanxitv.org/86)里有相關說明。
由上圖等值線分布可見,B6N6C6的硼和碳的價電子的費米穴分布范圍較廣,體現出電子有往較遠區域離域的能力,特別是其碳的費米穴分布特征和已知具有較顯著全局離域性的18碳環的很接近。相比之下,B9N9的硼和氮的費米穴分布廣度明顯不及B6N6C6的相應元素的,等值線只分布在距離參考點較近的原子上,體現出B9N9的價電子的離域能力相對較差。
上面的等值線圖適合定性討論,為了能把以上信息轉化成定量形式來更好地對比分析,文中定義了一個新的量叫做原子遠程離域指數(atomic remote delocalization index, ARDI)。離域性指數(DI)在《Multiwfn支持的分析化學鍵的方法一覽》(http://www.shanxitv.org/471)里專門介紹過,是衡量特定兩個原子間等效共享電子對數用的,而ARDI則是對單個原子定義的,定義為距離它兩個化學鍵以上的原子與當前原子的所有DI之和,顯然基于Multiwfn計算的所有原子間的DI可以簡單地手算出來各個原子的ARDI。ARDI體現了特定原子上的電子的遠程離域能力,數值越大說明遠程離域能力越強。環上各個原子上的電子有顯著的遠程離域能力顯然是這個環具備明顯芳香性的前提。各原子ARDI如下所示,可見B6N6C6上的原子的ARDI都較大,平均值與18碳環的碳的相仿佛,而B9N9的硼和氮的ARDI都遠小于B6N6C6相應元素的原子,這更充分體現了B9N9的電子的全局電子離域能力遠不如B6N6C6,進一步展現了B9N9芳香性遠低于B6N6C6的原因。

6 碳摻雜進B9N9使得芳香性巨幅提升的本質
根據前面的討論,文章已經充分證明了芳香性的關系是B6C6N6 ≈ C18 ? B9N9。為什么將碳元素摻雜進入無芳香性的B9N9,就能使其芳香性巨幅提升、電子全局離域能力大幅增加?實際上原因并不難理解。盡管前面的Mayer鍵級體現出B9N9中的硼-氮之間有一定π作用,但由于硼和氮的電負性差異非常大,這無疑會導致電子傾向于在氮上定域而難以往相鄰的硼上離域,至于往更遠的原子上離域就更是難上加難了。而碳的電負性則正好介于硼和氮之間,顯然硼-碳和氮-碳鍵的共價性顯著強于硼-氮鍵,因此電子更容易離域過去,相當于碳給硼和氮之間架設了能夠令電子容易離域過去的橋梁,充分調和了硼和氮之間電負性差異過大導致的電子跨鍵離域太難的矛盾。
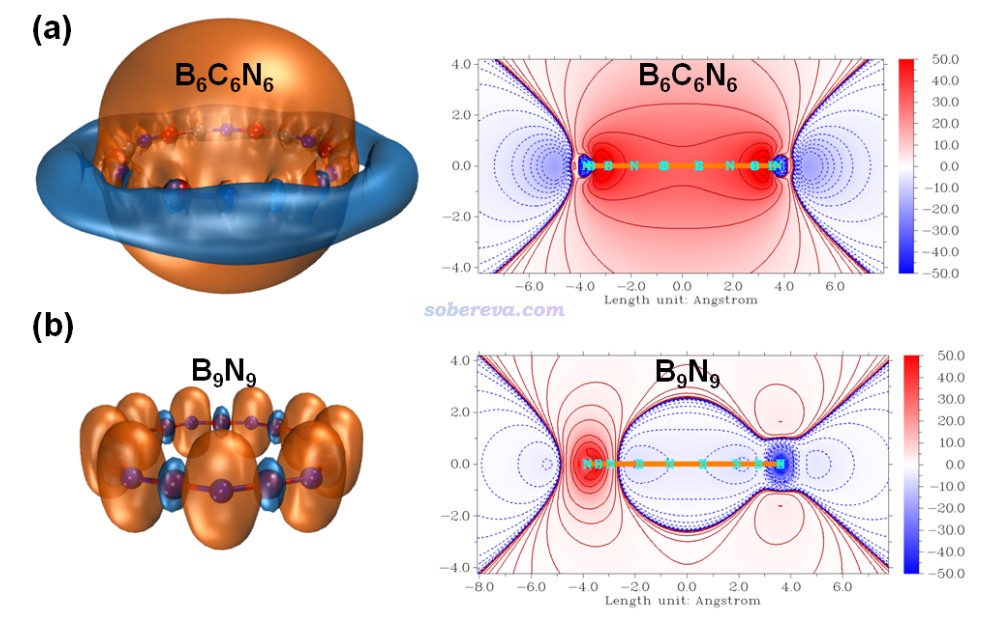
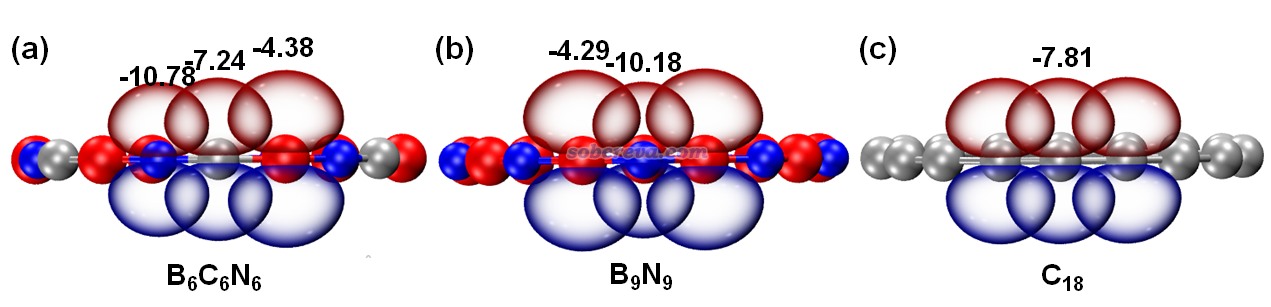
為了更好地展現碳原子起到的價值,文中將垂直于環的各原子的2p原子軌道繪制了出來,π-out電子正是分布在這些p軌道上,π-out分子軌道也正是它們線性組合產生的。具體來說,由于NBO程序產生的預自然原子軌道(PNAO)可以較合理地描述分子環境中的原子軌道,因此文中按照《使用Multiwfn繪制NBO及相關軌道》(http://www.shanxitv.org/134)中的做法,用Multiwfn結合VMD將相應的PNAO軌道用等值面圖形式做了展示,如下所示。等值面數值選為了適合對比的0.1。圖中也把PNAO的能量(eV)標注在了上面。由圖可清楚地看出,B9N9中的氮和硼的p軌道的空間延展程度差異很大,而且軌道能量差異也很大,勢必電子難以在它們之間離域。而從B6C6N6的圖中可見,碳的p軌道延展范圍和軌道能量都恰好介于硼和氮之間,它的引入無疑能夠顯著幫助電子離域過去。
7 B6N6C6異構體的芳香性
以上研究的那種以B-C-N作為重復單元排列的B6N6C6結構是否真的是B6N6C6的能量最低結構?是否B6N6C6化學組成的體系還有芳香性更強的異構體?這是個值得考察的有趣的問題。為此,文中還另外考慮了兩種B6N6C6構型,優化后得到的無虛頻結構如下所示,可見B6N6C6'構型讓所有碳盡可能連在一起,其它部分部分保持B9N9那樣硼-氮交錯的結構,而B6N6C6''構型則是由三個C-C-B-N-B-N重復單元構成的結構。這兩個結構的基態是閉殼層的,能量分別比前面研究的B-C-N為重復單元構成的B6N6C6結構能量低280.0和250.2 kcal/mol。這體現出B6N6C6中B-C和C-N的平均鍵能比B-N和C-C是要更小的。雖然前面研究的B6N6C6結構能量不是這種化學組成的能量最低結構,但并不意味著它是不穩定的。根據《使用ORCA做從頭算動力學(AIMD)的簡單例子》(http://www.shanxitv.org/576)中介紹的方法,文中對此體系做了較高溫度(500 K)下20 ps長度的基于wB97X-D3/6-311G*級別的從頭算分子動力學模擬,用VMD每1 ps繪制一次的結構疊加圖如下所示(顏色按照藍-白-紅變化),可見環狀結構始終很好維持著,并沒有發生異構化、解離等現象,體現出B6N6C6具有一定熱力學穩定性。

文中還對B6N6C6'和B6N6C6''構型計算了NICS(0)zz,發現分別僅有1.8和1.4 ppm,可見這倆構型沒有芳香性。這說明只有讓碳插入到硼和氮中間,從而以B-C-N作為重復單元時,碳才能真正起到顯著提升純硼-氮環體系的電子離域性的作用。
8 B6N6C6最低三重態激發態的芳香性
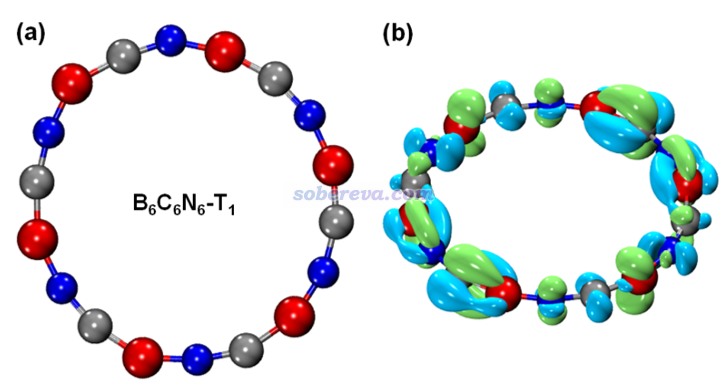
前面考察的都是B6N6C6的單重態基態(S0)。最后,文章還研究了B6N6C6最低三重態激發態(T1態)的芳香性。計算發現S0-T1垂直和絕熱激發能分別為1.62和1.17 eV。下圖(a)是優化后的T1態B6N6C6的結構,可見對稱性相對于S0態發生了下降。下圖(b)是按照《使用Multiwfn作電子密度差圖》(http://www.shanxitv.org/113)繪制的在B6N6C6的S0結構下T1態與S0態的密度差,藍色和綠色分別為電子密度降低和增加部分。由圖可見S0到T1態的電子激發是π-in → π-out的激發。由于垂直激發導致的電子密度變化破壞了B6N6C6基態的C6h對稱性,這也是為什么在T1態勢能面上從Franck-Condon點往極小點弛豫過程中幾何結構對稱性發生了下降。
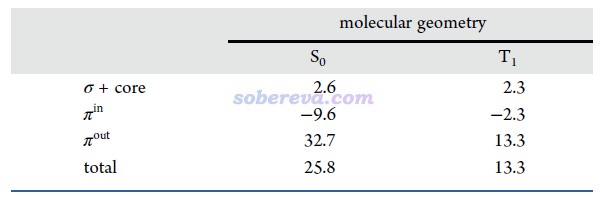
文中對B6N6C6的T1態計算了NICS(0)zz,并分解為了不同類型電子的貢獻。而且為了考察幾何結構弛豫的影響,在S0極小點和T1極小點分別做了這種計算,結果如下所示。可見無論在哪種結構下,T1態由于NICS(0)zz遠大于0,因此是顯著反芳香性的,這和Baird規則所述情況一致。Baird規則指出單重態基態滿足Hückel 4n+2規則的體系的最低三重態是反芳香性的。從NICS(0)zz還看到,T1態的反芳香性特征在T1極小點結構下比S0極小點結構下更弱,這體現出B6N6C6自發減小T1態的反芳香性可能是其S0→T1垂直激發后結構弛豫的重要驅動力。
9 總結
此文基于量子化學計算和Multiwfn等程序做波函數分析,非常全面系統研究了18碳環的等電子體B6N6C6的幾何結構和電子結構,從不同角度全面考察了其芳香性和電子離域性特征,并且和18碳環及其另外一個重要的等電子體B9N9進行了細致的對比。本文體現出以碳原子橋聯硼和氮原子,可以顯著提升純硼-氮體系的電子離域性。這不僅對電子結構產生重要影響,必然也同時會影響其它性質,諸如光學性質、電子輸運性質等。本文雖然研究的只是一維體系,可以預想電負性介于硼和氮之間的碳元素的摻雜必定也會對二維、三維純硼-氮體系產生顯著影響,而且摻雜方式的選取可能會對這些體系的性質起到一定調控作用。
如果讀者對于18碳環及其相關體系感興趣,非常推薦查看http://www.shanxitv.org/carbon_ring.html里匯總的本文作者的巨量其它研究工作,包含大量深入淺出的論文內容和研究思想介紹文章,以及對計算技術細節的附加說明。
]]>
百度網盤下載地址:https://pan.baidu.com/s/1ZfXjQbQp8zvFMwBs11F3uQ
提取碼:w6ri
這些文章的列表見本文末尾,或者看http://www.shanxitv.org/multiwfn/all_citation3.html里面的第13001~14000號文章。
這些文章可以視為Multiwfn的例子庫,便于用戶了解Multiwfn在量子化學實際研究中能發揮的重要價值。如果想找某一類應用實例,可以利用Acrobat對指定文件夾里全部文檔進行搜索的功能搜索相關關鍵詞。
由于本文件包里的文章都是它們剛在期刊網站上刊登的時候我就下載的,所以文檔中大多沒有頁碼、卷號,且多數都是還沒經過proofing的原始狀態,大家只要到google學術搜索里搜索標題就能找到文章最終版鏈接。在Multiwfn主頁(http://www.shanxitv.org/multiwfn)的Cited by頁面里也有所有引用了Multiwfn的文章的匯總列表和文章鏈接。
之前的引用Multiwfn的第1~1010篇文章可以從這里下載:http://bbs.keinsci.com/thread-2850-1-1.html
之前的引用Multiwfn的第1011~2001篇文章可以從這里下載:http://bbs.keinsci.com/thread-6754-1-1.html
之前的引用Multiwfn的第2002~3000篇文章可以從這里下載:http://bbs.keinsci.com/thread-11023-1-1.html
之前的引用Multiwfn的第3001~4000篇文章可以從這里下載:http://bbs.keinsci.com/thread-14181-1-1.html
之前的引用Multiwfn的第4001~5000篇文章可以從這里下載:http://bbs.keinsci.com/thread-16840-1-1.html
之前的引用Multiwfn的第5001~6000篇文章可以從這里下載:http://bbs.keinsci.com/thread-19818-1-1.html
之前的引用Multiwfn的第6001~7000篇文章可以從這里下載:http://bbs.keinsci.com/thread-22597-1-1.html
之前的引用Multiwfn的第7001~8000篇文章可以從這里下載:http://bbs.keinsci.com/thread-25497-1-1.html
之前的引用Multiwfn的第8001~9000篇文章可以從這里下載:http://bbs.keinsci.com/thread-27978-1-1.html
之前的引用Multiwfn的第9001~10000篇文章可以從這里下載:http://bbs.keinsci.com/thread-31224-1-1.html
之前的引用Multiwfn的第10001~11000篇文章可以從這里下載:http://bbs.keinsci.com/thread-34124-1-1.html
之前的引用Multiwfn的第11001~12000篇文章可以從這里下載:http://bbs.keinsci.com/thread-36387-1-1.html
之前的引用Multiwfn的第12001~13000篇文章可以從這里下載:http://bbs.keinsci.com/thread-38881-1-1.html
再次順帶強調關于恰當引用的問題。在Multiwfn程序主頁的首頁和Download頁面、程序手冊、程序啟動時屏幕上顯示的信息、程序包內的諸多文件(Multiwfn quick start.pdf文檔、How to cite Multiwfn.pdf文檔、LICENSE.txt文件)、Multiwfn入門貼(http://www.shanxitv.org/167)、Multiwfn FAQ(http://www.shanxitv.org/452)等盡可能多、用戶可以輕易看到的所有地方都非常非常非常明確強調如何對Multiwfn進行正確的引用,但使用Multiwfn的文章中不引用或者胡亂引用的現象依然十分嚴重(特別是中國的文章尤甚)!第13001~14000篇文章里沒按要求恰當引用Multiwfn的文章多達165篇!經常是只提及Multiwfn但不引用,甚至在引用Multiwfn的地方引用的卻是和Multiwfn根本沒有任何直接聯系的其他人的文章。還有不少文章里作者明明在研究中用了Multiwfn做分析,在論文里竟然連Multiwfn的名字都不提。不按明確聲明的方式恰當引用,對免費而且沒有任何經費支持的程序的發展十分不利,同時也是嚴重缺乏學術道德的行為。借此機會再次強烈呼吁用戶重視對Multiwfn的恰當引用。最恰當的引用方式見Multiwfn可執行文件包內的How to cite Multiwfn.pdf文檔的說明。沒有恰當引用Multiwfn的文章在本次的文件包里的full list.txt里都已經注上了Multiwfn was not properly cited!的字樣,在此予以強烈批評。
文章列表如下(有的文章標題太長,為了避免文件路徑超過Windows限制,只保留了前面一部分)
2-(Indol-3-yl)- and 6-(pyrrol-2-yl)-substituted (bi)pyridine-based AIE-probes-fluorophores-synthesis and photophysical studies
2-D polypseudo-rotaxane network in (4Z,N'Z)-N’-(amino(pyridine-2-yl)methyl)isonicotionhydrazonic (apminh) Pb(II) complex-Synthesis, Structural and Quantum computational studies
A 3,5-dinitropyridin-2yl Substituted Flavonol-based Fluorescent Probe for Rapid Detection of H2S in Water, Foodstuff Samples and Living Cells
A C-PCM investigation of the linear and nonlinear optical properties in Cr(CO)3(η6-corannulene) complex
A Class of Activatable NIR-II Photoacoustic Dyes for High-Contrast Bioimaging
A combined experimental and theoretical study on the structures, dynamics and interactions of BMIMTfO and BMIMTFSI binary ionic liquid mixtures
A Comparison Study of Roseolumiflavin Solvates Structural and Energetic Perspective on Their Stability
A computational insight into enhancement of photovoltaic properties of non-fullerene acceptors by end-group modulations in the structural framework of INPIC molecule
A computational study on developing Tween Type-Graphdiyne based fluorescent sensor for rapid and sensitive detection of vitamin C
A computational study on the coordination modes and electron absorption spectra of the complexes U(IV) with N,N,N′,N′-tetramethyl-diglycolamide and anions
A cyclic deep-red bis-squaraine molecular scaffold for improving random laser performance
A DFT investigation of the catalytic oxidation of benzyl alcohol using graphene oxide
A DFT study for improving the photovoltaic performance of organic solar cells by designing symmetric non-fullerene acceptors by quantum chemical modification on pre-existed LC81 molecule
A DFT Study of NHC-Catalyzed Reaction of [3 + 3] Annulation of 2-Bromoenals and Thioamides
A fast ionic transport copolymeric network for stable quasi-solid lithium metal battery
A Gatekeeper Residue Controls Aromatic Acceptor Specificity of the PHB-Type UbiA Prenyltransferases
A Ground-State Dual-Descriptor Strategy for Screening Efficient Singlet Fission Systems
A Highly NIR Emissive Cu16Pd1 Nanocluster
A lithium cycloparaphenylene as an emerging second-order non-linear optical molecular switch
A Metal-Free Molecular Ferroelectric [4-Me-cyclohexylamine]ClO4 Introduced by Boat and Chair Conformations of Cyclohexylamine
A more flexible long-chain bis(salamo)-like fluorescent probe based on FRET and ICT effects for the identification of aluminum ion in the environment and plants
A Multi-Component Nano-Co-Delivery System Utilizing Astragalus Polysaccharides as Carriers for Improving Biopharmaceutical Properties of Astragalus Flavonoids
A new steroid with potent antimicrobial activities and two new polyketides from Penicillium variabile EN-394, a fungus obtained from the marine red alga Rhodomela confervoides
A new turn-on fluorescent probe for fast detection of diabetic biomarker beta-hydroxybutyrate in vitro
A novel dopamine electrochemical sensor based on multiwall carbon nanotubes-cetyltrimethylammonium bromide-nitrogen doped ultra-thin carbon nanosheets composites modified glassy carbon electrode
A novel fused bi-macrocyclic host for sensitive detection of Cr2O72? based on enrichment effect
A novel intramolecular charge transfer-based near-infrared fluorescent probe with large Stokes shift for highly sensitive detection of cysteine in vivo
A Potential Nonlinear Optical Material for Crown-Porphyrin Complexes-Alkali Metal Doping and Transition Metal Introducing
A quantum chemical and molecular dynamics simulation study on photo-oxidative aging of polyethylene
A Quantum Chemical Study-Thoughtful Exploration for Optimal Donors in Y-Type Dual Donor-Based Dye Sensitizers
A Rational Design of Alkali Metal-Doped Germanium Carbide Nanoclusters for High Nonlinear Optical Response and Ultraviolet Transparency
A review of ultra-high temperature heat-resistant energetic materials
A Spot Ionogel for Visual Early-Warning of Illegal Lead Concentrations Coupling Inorganic Perovskite Crystallization and Photoluminescence
A study on the preparation of V2O5 from NH4V3O8 based on thermal decomposition including the thermal decomposition temperature effect, mechanism and kinetics
A Theoretical Molecular Design of Acridine-Anthracene-Polyoxometalate Hybrid Materials for Enhanced Dye-Sensitized Solar Cells Performance
A theoretical study on hydrated sodium ion-phenylalanine clusters Na+(Phe)(H2O)n (n = 0-6; Phe = phenylalanine)
A theoretical study on π-stacking and ferromagnetism of perylene diimide radical anion dimer
A thermal-responsive hydrogel to capture uranium(VI) from aqueous solution-properties and mechanism
A-D-A Type Nonfullerene Acceptors Synthesized by Core Segmentation and Isomerization for Realizing Organic Solar Cells with Low Nonradiative Energy Loss
Absorption characteristics, model, and molecular mechanism of hydrogen sulfide in morpholine acetate aqueous solution
Absorption studies on serotonin neurotransmitter with the platinum metal cluster using the gas phase and different solvents, topological and non-covalent interaction
Accelerated degradation and toxicity reduction of ribavirin by organic free radicals in the ferrate-acetylacetonate system
Accelerated ion transportation in liquid crystalline polymer networks for superior solid-state lithium metal batteries
Accelerating the concerted double proton transfer process of 2,2′-bipyridine-3,3′-diol-5,5′-dicarboxylate acid ethyl ester (BPDC) molecule by centrosymmetric dual intermolecular hydrogen bonds
Acceptor engineering for modulating circularly polarized luminescence and thermally activated delayed fluorescence properties
Accounting for super-, plateau- and mesa-rate burning by lead and copper-based ballistic modifiers in double-base propellants-a computational study
Accurate and Efficient Prediction of Post-Hartree–Fock Polarizabilities of Condensed-Phase Systems
Achieving narrowband emissions with tunable colors for multiple resonance-thermally activated delayed fluorescence materials-Effect of Boron-Nitrogen number and position
Achieving TADF and RTP with Stimulus-Responsiveness and Tunability from Phenothiazine-Based Donor?Acceptor Molecules
Achieving the cooperative and stepwise regulation of ESIDPT process in AFBD by introducing different electronic groups
Activation Model and Origins of Selectivity for Chiral Phosphoric Acid Catalyzed Diradical Reactions
Activation of H2 Molecules on Platinum and Platinum–Vanadium Clusters-DFT Quantum Chemical Modeling
Acylhydrazone functionalized naphthalene-based self-assembled supramolecular gels for efficient fluorescence detection of Fe3+
Additional O doping significantly improved the catalytic performance of Mn-O co-doped g-C3N4
Adsorption and diffusion of actinyls on the basal gibbsite (001) surface-A theoretical perspective
Adsorption behavior, different green solvent effect and surface enhanced Raman spectra (SERS) investigation on inhibition of SARS-CoV-2 by antineoplastic drug Carmofur
Adsorption dynamics of benzene derivatives onto the surface of hydroxylated silica upon photoexcitation
Adsorption mechanism of amino acid ionic liquids on the N-doped graphene surface for electrochemical double layer capacitors-A density functional theory study
Adsorption of Choline Phenylalanilate on Polyaromatic Hydrocarbon-Shaped Graphene and Reaction Mechanism with CO2
Adsorption of Multi-Collector on Long-Flame Coal Surface via Density Functional Theory Calculation and Molecular Dynamics Simulation
Adsorption studies of dye molecule on two-dimensional assembly of porphyrin using density functional theory
Alcohol regulated phase change absorbent for efficient carbon dioxide capture-Mechanism and energy consumption
Alkali-metal coating-an effective method to inject electrons into cage molecules, and achieve direct metal-metal bonds and spherical aromaticity for endohedral metallofullerenes
Alkali-thermal activated persulfate treatment of tetrabromobisphenol a in soil-Parameter optimization, mechanism, degradation pathway and toxicity evaluation
Alkoxy substitution on simple non-fused electron acceptors for tuning the photoelectric properties of organic solar cells
Amphiphilic heteropolyacid-based sulfonic acid-functionalised ionic liquids as efficient catalysts for biodiesel production
An alkylation route for developing novel liquid aviation fuels from α-pinene and mixed isobutane-isobutene
An effective HBT-indanone-based fluorescent probe for cysteine detection in cells
An effective strategy to design high-performance wide bandgap polymer donors for organic solar cells by increasing the backbone curvature
An Extensive Experimental and DFT Studies on Highly Selective Detection of Nitrobenzene Through Deferasirox Based New Fluorescent Sensor
An in silico investigation of Kv2.1 potassium channel-Model building and inhibitors binding sites analysis
An in-situ electrogenerated persulfate and its activation by functionalized sludge biochar for efficient degradation of sulfamethoxazole
An insight into the interaction of silicon decorated fullerene and single walled carbon nanotube towards umifenovir
An intrinsic safe siloxane ether-based electrolyte for lithium-sulfur batteries at high temperatures
An investigation on spectroscopic, wavefunction dependent reactivity, docking and anti-Covid-19 ability of Flupentixol dihydrochloride
An oxazole-derived Schiff base as a multifunctional fluorescence sensor towards Al3+ and Ga3+ in different media
ANALYSES ON THE REACTIVITY, TOPOLOGY AND BIOACTIVITY OF FLUAZINAM USING DENSITY FUNCTIONAL THEORY
Anion optimization for bifunctional surface passivation in perovskite solar cells
Antibacterial and osteogenic activities of thiolated and aminated yttria-stabilized tetragonal zirconia polycrystal with tolerance to low temperature degradation
Antimicrobial activity prediction, inter- and intramolecular charge transfer investigation, reactivity analysis and molecular docking studies of adenine derivatives
Application of Computational Chemistry in the Teaching of the Regioselectivity Mechanism of DA Reaction
Applied and Computational Investigation of Plasticizing Effects of Dibutyl Maleate on Polymethyl Methacrylate Acrylic Resin Material
Array-Based Clusters of Copper with Largely Exposed Metal Sites for Promoting Catalysis
Asepterpenedol A, a novel indole sesquiterpene with a rare 7,6,5,5,6,6-hexacyclic scaffold from a marine-derived fungus Chloridium sp. NBU3282
Assembling Nitroamino and Amino Groups on a Pyrazole-1,3,4-Oxadiazole Framework for the Construction of High-Performance and Insensitive Energetic Materials
Assessing cationic dye adsorption mechanisms on MIL-53 (Al) nanostructured MOF materials using quantum chemical and molecular simulations
Assessing the dipolar-octupolar NLO behavior of substituted thiosemicarbazone assemblies
Assessments of carbon and boron nitride graphdiyne nanosheets for exploring the amphetamine drug adsorbents-sensors along with density functional theory
Atmospheric oxidation of (E)-β-farnesene initiated by hydroxyl radical-New formation mechanism of HOMs
Atomic electronegativity-dependent intramolecular hydrogen bond and fluorescence characteristics of novel scaffold-based fluorophore-a TD-DFT study
Atomic scale microparameter analysis of modified natural ester molecules related to impulse discharge characteristics under electric field
Au nanoparticle-decorated Ni2P nanosheet arrays with porous grids for the determination of hydroquinone in domestic sewage
Azo-benzoic acid derivatives directed dinuclear and tetranuclear association of trimethyltin(IV) complex components and their biological activities
B-N B-O Contained Heterocycles as Fusion Locker in Multi-Resonance Frameworks towards Highly-efficient and Stable Ultra-Narrowband Emission
Benchmark computational investigations for the basic model of the salt-water complex-NaCl(H2O) and its anion NaCl(H2O)?
Benzodithiophene-based Acceptor-Donor-Acceptor-type compounds for highly efficient organic photovoltaic cells
Benzotiyazolden Elde Edilen Baz? Azo Boyarmaddelerin Sentezi ve Yo?unluk Fonksiyonel Teorisi ile Analizi
Bidirectional Charge Transfer at the Heme Iron in Reversible and Quasi-irreversible Cytochrome P450 Inhibition
Biobased Biodegradable Copolyesters from 2,5-Thiophenedicarboxylic Acid-Effect of Aliphatic Diols on Barrier Properties and Degradation
Biochars from modified sugarcane bagasse for manganese removal from mining effluents
Biodegradability analysis of Dioxins through in silico methods-Model construction and mechanism analysis
Biological noncovalent NO...V interactions insights from theory and protein data bank analyses
Boosting Photocatalytic Hydrogen Evolution Through Local Charge Polarization in Chemically Bonded Single-Molecule Junctions Between Ketone Molecules and Covalent Organic Frameworks
Boron-based Pd3B26 alloy cluster as a nanoscale antifriction bearing system
Broadband Terahertz Spectroscopy and Weak Interactions of Adenosine with Vibrational Mode Analysis
Br?nsted ionic liquids as catalysts for synthesis of 2-pyrazoline derivatives from ketazines
Calculation and Analysis of Electron Transition Spectra, in Computational Simulation in Nanophotonics and Spectroscopy
Calculation of excited states of monolayer TPPA-COF based on First-principles
Calculation of Nonlinear Optical Properties
Can Catenated Nitrogen Compounds with Amine-like Structures Become Candidates for High-Energy-Density Compounds
Carbon Bond Catalysis-Dialkyl Sulfates, Alkyl Sulfonates and Alkyl Hal-ides as Catalysts in Acetal Forming and Related Reactions
Carbon nanobelts with zigzag and armchair edge and interlocked carbon nanobelts for chirality
Carbonized polymer dots assisted thermally activated delayed fluorescence to achieve stronger luminescence
Carboxymethyl cellulose-based supramolecular hydrogel with thermo-responsive gel-sol transition for temporary plugging of oil pipeline in hot work
Catalytic Enantioselective Nucleophilic Addition to Arynes by a New Quaternary Guanidinium Salt-Based Phase-Transfer Catalyst
Catalytic Hydrogenation of Nitroarenes over Ag33 Nanoclusters-The Ligand Effect
Cellulose dissolution and regeneration behavior via DBU-levulinic acid solvents
Cg…Cg interactions driven 1D polymeric chains bridged by lattice solvents in N3-(2-pyridoyl)-4-pyridinecarboxamidrazone Pb(II) complex
Characterization, Analysis, and Theoretical Calculation Studies of Solvates and Cocrystals of Betulin
Charge transfer in superbase n-type doping of PCBM induced by deprotonation
Chemistry of Cyclo-[2n]-Carbon-A Many-Particle Quantum Mechanics Investigation
Chiral coherent control of electronic population transfer-towards all-optical and highly enantioselective photochemistry
Chiral Substitution on Spaced Cations Lead to Improved Properties and Reversible Phase Transition, Broadband Emission in Parent Compound (3APr)PbBr4
Cocrystal of 4,5′-dinitro-3′H-2,4′-bi(1,2,3-triazole) and imidazole-a promising strategy for development of heat-resistant energetic materials
COCRYSTAL PREDICTION OF THE SALICYLIC ACIDNICOTINAMIDE
Cocrystals of Favipiravir-Improved Physicochemical Properties and Solution Stability Study
Combined FTIR-Raman spectroscopic studies and ab initio electronic structure calculations of Dithiothreitol
Combined Molecular Dynamics and Coordinate Driving Method for Automatically Searching Complicated Reaction Pathways
Comparative analysis of interaction of ionic liquids with porous and non-porous electrode materials
Comparative Study of the Optical and Electronic Properties of Y6 Derivatives-A Theoretical Study
Comparative study on ionic liquids and deep eutectic solvents in the separation of fuel additive isopropyl alcohol and ethyl acetate by the experimental study and molecular simulation
Comparison of reactive sites of different conjugated linoleic acid molecules by quantum chemical calculations
Complexes of the noble-gas atoms with borazine-Theoretical insights into structure, stability, and bonding character
Comprehensive protection of Zn metal anode via caprolactam towards highly stable aqueous zinc ions batteries
Computational analyses of cooperativity between pnicogen and halogen bonds in H2FP-pyrimidine-ClF complex
Computational Analysis on Molecular Stability and Binding Affinity of 3-(Aminothiazolyl)Quinolone Derivative as Multitargeting Antibacterial Agents
Computational and Experimental Study on the Mechanism of CO2 Production during Photo-Oxidative Degradation of Poly(butylene adipate-co-terephthalate)
Computational Chemistry and Molecular Modeling Techniques for the Study of Micropeptin EI-964
Computational investigation of the structures, properties, and host-guest chemistry of prism[n]arenes
Computational Manifestation of Nitro-Substituted Tris(triazole)-Understanding the Impact of Isomerism on Performance-Stability Parameters
Computational screening of metalloporphyrin-based drug carriers for antitumor drug 5-fluorouracil
Computational studies into the chemical nature, thermal behaviour, solvent role, reactivity and biological evaluation of Rigidin E
Computational Study of the Photophysical Properties and Electronic Structure of Iridium(III) Photosensitizers Complexes with Electron-withdrawing Groups
Computational study on alkali and alkaline earth metal decorated B20 cluster for hydrogen storage application
Computational Study on Piperazine-1,4-Diium Acetate Using Dft Investigations-Structural Aspects, Topological and Nonlinear Optical Properties
Computational Study on the Mechanisms of Inhibition of SARS-CoV-2 Mpro by Aldehyde Warhead Based on DFT
Computational Study on the Route of Cooperative Organocatalysis Utilizing Thiourea and Halogen Bond Donor Mixture
Computational, Reactivity, Fukui Function, Molecular Docking, and Spectroscopic Studies of a Novel (E)-1-Benzyl-3-(2-(Pyrindin-2-yl)Hydrazono)Indolin-2-One
Concerted Intramolecular and Intermolecular Charge Transfer for High-Efficiency Near-Infrared Thermally Activated Delayed Fluorescent Materials Approaching 900 nm
Concretized structural evolution supported assembly-controlled film-forming kinetics in slot-die coated organic photovoltaics
Conjugated Coupler Curvature Enhances Magnetic Spin Coupling in π-Diradicals
Construction of intramolecular donor–acceptor type carbon nitride for photocatalytic hydrogen production
Construction of supramolecular S-scheme heterojunctions assisted by hydrogen bond subtle-tuning actuates highly efficient photocatalytic oxidation
Controllable fluorescent switch based on ferrocene derivatives with AIE-active tetraphenylethene moiety
Controlling the CO2 Reduction Reaction through Dual-Atom Catalysts Embedded in Expanded Porphyrins
Controlling the Energetic Properties of N-Methylene-C-Linked 4-Hydroxy-3,5-dinitropyrazole- and Tetrazole-Based Compounds via a Selective Mono- and Dicationic Salt Formation Strategy
Cooperativity between H-Bonds and Tetrel Bonds. Transformation of a Noncovalent C···N Tetrel Bond to a Covalent Bond
Coordination-induced bond weakening in NiC3-An experimental and theoretical investigation
Copper-Catalyzed Reaction of N-Monosubstituted Hydrazones with CBr4-Unexpected Fragmentation and Mechanistic Study
Copper–zinc-chitosan complex hydrogels-Rheological, degradation and biological properties
Correlating the Solvating Power of Solvents with the Strength of Ion-Dipole Interaction in Electrolytes of Lithium-ion Batteries
Corrosion protection performance of Laurhydrazide N′-propan-3-one (LHP) adsorbed on zinc surface-A DFT-MD simulation investigation
Counter-Anions Rendered Weak-Interactions Perturb the Stability of Tyrosinase-Mimicked Peroxo-Dicopper(II) Active Site
Covalent Organic Framework Membrane with Angstrom Discrimination in Pore Size for Highly Permselective Ionic Liquid Nanofiltration
Covalent organic framework-based magnetic solid-phase extraction coupled with gas chromatography-tandem mass spectrometry for the determination of trace phthalate esters in liquid foods
Crystal growth, HOMO-LUMO engineering and charge-transfer emission of TCNB-based donor-acceptor cocrystals
Crystal structure, supramolecular assembly exploration by Hirshfeld surface analysis and DFT inspection of the synthesized functionalized crystalline anilide
Crystallographic, theoretical and conductivity studies of two new complexes [Ni(II) and Cu(II)] based on mixed ligands approach
Cycloaddition – Retro-Electrocyclization Click Reaction of Amine End-Capped Oligoynes with Tetracyanoethylene
C≡N Stretching Frequency as a Convenient Reporter of Charge Separation in Molecular Systems
Data-Driven Insights into the Fluorescence of Asphaltene Aggregates Using Extended Frenkel Exciton Model
Dauricine interferes with SARS-CoV-2 variants infection by blocking the interface between RBD and ACE2
Deciphering melting behaviors of energetic compounds using interpretable Machine learning for melt-castable applications
Deciphering the electrochemical sensing capability of novel Ga12As12 nanocluster towards chemical warfare phosgene gas
Deciphering the Mechanistic Insights of Temporary Directing-Group-Assisted meta-Alkenylation of Complex Biaryl Systems
Decoding Chemical Bonds-Assessment of the Basis Set Effect on Overlap Electron Density Descriptors and Topological Properties in Comparison to QTAIM
Decorating Mg12O12 Nanocage with Late First-Row Transition Metals To Act as Single-Atom Catalysts for the Hydrogen Evolution Reaction
Deep eutectic solvents for effective removal of indoor formaldehyde-Theoretical design, experiment, and adsorption mechanism study
Deep removal of dichloromethane using ionic liquids-Thermodynamic and molecular insights
Defect-free PEO membrane fabrication by hydrogen bonding coupling thermal annealing for carbon capture
Degradation Chemistry of Dimetallaboranes, [(Cp M)2B5H9+x] with [Ph2Se2] (x = 2 or 0, M = V or Cr)
Degradation of xanthene-based dyes by photoactivated persulfate-experimental and computational studies
Dehydration of gypsum waste to recyclable anhydrite using a nano-film reservoir under ambient conditions
Density functional theory studies on properties of cluster ConMoS (n=1 ~ 5)
Dependence of Intramolecular Hydrogen Bond on Conformational Flexibility in Linear Aminoalcohols
DESENHO DE NOVOS HETERODíMEROS COMO PROPOSTA à INIBIDORES DA ACHE HUMANA
Design and synthesis of intrinsic black polyimide with full visible-light absorption and low coefficient of thermal expansion for black flexible copper clad laminates
Design of 3D Metal–Organic Material with Multiple Redox-Active Sites for High-Performance Lithium-Ion Batteries
Design of electron-donating group substituted 2-PAM analogs as antidotes for organophosphate insecticide poisoning
Design of insensitive high explosives based on FOX-7-a theoretical prospectives
Design, Synthesis and Characterization of Ferrocene Containing Aromatic Foldamers & Synthesis and Study of Novel Indole Based Chalcones for Anti-cancer Activities
Design, synthesis, and properties of soluble intrinsic black polyimide bearing tetraphenylcyclopentadienone units
Designing Benzodithiophene-Based Small Molecule Donors for Organic Solar Cells by Regulation of Halogenation Effects
Designing Nanoarchitecture of NiCu Dealloyed Nanoparticles on Hierarchical Co Nanosheets for Alkaline Overall Water Splitting at Low Cell Voltage
Designing of banana shaped chromophores via molecular engineering of terminal groups to probe photovoltaic behavior of organic solar cell materials
Designing polyurethane-based microcapsules with tailored swelling behaviours for enhanced oil recovery
Desulfurization of diesel via joint adsorption and extraction using a porous liquid derived from ZIF-8 and a phosphonium-type ionic liquid
Detonation properties and sensitivities of a series of -NHNH- -N?=?N- bridged tetrazole-based energetic materials
Development of a fluorescent sensor based on TPE-Fc and GSH-AuNCs for the detection of organophosphorus pesticide residues in vegetables
DFT analysis of the adsorption of bisphenol A (BPA) on pristine and oxidized phosphorene
DFT computational and spectroscopic studies on andrographolide from different solvent effect
DFT Model of Elemental Sulfur in Sulfolane Solutions
DFT studies of solvent effect in hydrogen abstraction reactions from different allyl-type monomers with benzoyl radical
DFT Studies on Pd-Xiao-Phos-Catalyzed Asymmetric Arylation of Secondary Phosphine Oxides
DFT Studies on the Effect of Additives on Stereoselectivity in the Polymerization of Styrene Catalyzed by Rare Earth Metal Complexes
DFT study of N-modified graphene-loaded monometallic cobalt for acetylene hydrochlorination reaction
DFT study on adsorption of small gas molecules on Pt-capped armchair and zigzag single-walled carbon nanotubes
DFT study on the intermolecular interaction between two energetic ionic salts-a case of TKX-50 and ADN
DFT, ADME studies and evaluation of the binding with HSA and MAO-B inhibitory potential of protoberberine alkaloids from Guatteria friesiana
DFT-Based Study of the Structure, Stability, and Spectral and Optical Properties of Gas-Phase NbMgn (n = 2–12) Clusters
DFT-based study on the differences between odd and even Cn (n = 6–31) ring clusters
Difference in the complexation of cholesterol with β-cyclodextrin derivatives-A combined theoretical and experimental study
Dihydroxanthene-derived fluorescent probe with near-infrared excitation and emission maxima for detecting human carboxylesterase-2 and bioimaging
Dimeric cation-cation aggregates stabilized by 2Ch-2N chalcogen bonds-crystallographic and theoretical evidences
Dipole Moment of the S0 → S1 Chlorophyll a Transition in Solvents with a Varied Refraction Index
Direct time-resolved observation of surface-bound carbon dioxide radical anions on metallic nanocatalysts
Directionality and additivity effects of molecular acidity and aromaticity for substituted benzoic acids under external electric fields
Discovery of 2,4-diphenyl-substituted thiazole derivatives as PRMT1 inhibitors and investigation of their anti-cervical cancer effects
Discovery of a series of silicon-based ferrimagnets in CrMnSin (n?=?4–20) clusters
Discovery of natural multi-targets neuraminidase inhibitor glycosides compounds against influenza A virus through network pharmacology, virtual screening, molecular dynamics simulation, and in vitro experiment
Discovery of potential RIPK1 inhibitors by machine learning and molecular dynamics simulations
Discovery of the Caged-Vibsane Norditerpenoids with Unprecedented Chemical Architectures and Exploration of Their Various Acid Tolerances
Distinct Electronic Effect on Borane and Boryl Nickel Complexes for Catalyzing H2 Activation
Distortion-controlled 1,2-dicarbene reactivity of 3-triflyloxybenzynes-a theoretical approach
Donor Radii in Rare-Earth Complexes
Dose alkyl chain unsaturation could affect tunability of the aryl alkyl imidazolium-based ion pairs
Double-chain conjugated carbonyl polymer cathode for rechargeable magnesium batteries
Dual-emissive Iridium(III) Complexes as Phosphorescent Probes with Orthogonal Responses to Analyte Binding and Oxygen Quenching
Dual-state emission of D-A-D type benzothiadiazole derivatives for the sensitive detection of amine compounds
Dye Modified Phenylenediamine Oligomers-Theoretical Studies on Drug Binding for Their Potential Application in Drug Sensors
Dynamic Protonation States Underlie Carbene Formation in ThDP-Dependent Enzymes-A Theoretical Study
Effect of anion and methylation on the interionic interactions and reactivity of 1-butyl-2,3-dimethyl imidazolium-based ionic liquids
Effect of conjugation length on fluorescence characteristics of carbon dots
Effect of coordination environment on polycondensation of polyester catalyzed by organic titanium compounds
Effect of electric field intensity on droplet fragmentation in oil-in-water-in-oil (O-W-O) emulsions-A molecular dynamics study
Effect of Functional Group Position in Co-formers and Solvent on Cocrystal Polymorphism-Stoichiomorphism-A Case Study
Effect of ligand coordination on the mechanism and regioselectivity of cobalt-catalyzed hydroboration-cyclization of 1,6-enynes
Effect of microplastics on the adherence of coexisting background organic contaminants to natural organic matter in water
Effect of microplastics on the degradation of tetracycline in a soil microbial electric field
Effect of push–Pull functional group on the ESIPT behaviour and photophysical property of thiolflavone-based compound-a TD-DFT investigation
Effect of solvents on intra- and inter-molecular interactions of oligothiophenes
Effect of steric hindrance and number of substituents on transfer and interface properties of Y-shaped hole-transporting materials for perovskite solar cells
Effect of tailoring π-linkers with extended conjugation on the SJ-IC molecule for achieving high VOC and improved charge mobility towards enhanced photovoltaic applications
Effect of thionation of carbonyl group on the photophysics of compact spiro rhodamine-naphthalimide electron donor-acceptor dyads
Effective electrostatic potential surface and aromatic affinity as quantitative guide for deep extraction denitrification from aromatic-rich oils
Effects of Au6 and Au20 Adsorption Sites of Cyromazine–Au Complexes by Raman Spectroscopy and Density Functional Theory
Effects of external electric fields on molecular properties of nitrogen-tetrafluoromethane complex-A density functional theory study
Efficient absorption of low pressure NH3 by non-ionic phenol-based deep eutectic solvents with multiple acidic sites
Efficient adsorption of bisphenol A by metal-organic frameworks-derived N-doped carbon nanoflakes-adsorption performance and mechanism investigation
Efficient and Selective Recovery of Gold from Thiosulfate Leaching Solution Using Functionalized β-Cyclodextrins Synthesized by a Steric Hindrance Strategy
Efficient capture of benzene and its homologues volatile organic compounds with protic [MIM][NTF2] and aprotic [EMIM][NTF2] ionic liquids
Efforts to understand the degree of carboxylate substitution in cis-[Pt(S(O)Me2)(guanidine)Cl2]-Synthetic, Structural and DFT Studies
Electric-Field Effects on the Internal Charge Reorganization Energies of Crystalline Organic Semiconductors
Electrical Properties of CF3SO2F Insulating Gas based on Density Functional Theory
Electrically Controlled Adsorptive Membranes with Tunable Affinity for Selective Chromium (VI) Separation from Water
Electrochemical properties of endohedral halide (F-, Cl- and Br-) encapsulated Mg12O12 nanocage for metal-ion batteries
Electron Density Learning of Z-Bonds in Ionic Liquids and Its Application
Electron density-based protocol to recover the interacting quantum atoms components of intermolecular binding energy
Electronegativity effect on the ESIPT process of 4′-N,N-dimethylamino-3-hydroxyflavone (DMA3HF) and its derivatives
Electronic and gas sensing properties of ultrathin TiO2 quantum dots-A first-principles study
Electronic and optical properties of chemically modified 2D GaAs nanoribbons
Electronic and Optical Properties of Finite Gallium Sulfide Nano Ribbons-A First-Principles Study
Electronic delocalization in charged macrocycles is associated with global aromaticity
Electronic Modulation Induced Luminescence from Triphenylamine Derivative and Temperature Sensor Application
Electronic Rearrangement in Steps of Reductive Elimination of Polar Electrophiles Leads to Refinement of Redox Events
Elucidating the electronic synergetic effects in heteroatomic doped FeN4-C-N-R (R= -F, -Cl, -Br) oxygen reduction catalysts
Elucidating the mechanism and regioselectivity of phosphine-catalyzed transformation of MBH carbonate
Elucidation of collagen amino acid interactions with metals (B, Ni) encapsulated graphene-PEDOT material
End-capped engineering of carbazole based dopant free hole transporting material with improved power conversion efficiency
Energetic Bi-diazole 'Transformers' toward High Energy Thermostable Energetic Compounds
Energetic N-Nitramino-Substituted 1,2,4-Triazole and Corresponding Salts-Green Primary Explosives with Excellent Detonation Performance
Energetic performance of trinitromethyl nitrotriazole (TNMNT) and its energetic salts
Energy Barrier of Photoinduced Charge Separation in the Reaction Centers of Photosystems I and II
Engineering Diselenide-IR780 Homodimeric Nanoassemblies with Enhanced Photodynamic and Immunotherapeutic Effects for Triple-Negative Breast Cancer Treatment
Engineering Radioactive Microspheres for Intra-Arterial Brachytherapy Using Radiation-Induced Graft Polymerization
Engineering singlet and triplet excitons of TADF emitters by different host-guest interactions
Enhanced blackness and solubility of polyimide via introducing propeller-shaped chromophore
Enhanced electrochemical treatment of humic acids and metal ions in leachate concentrate
Enhanced TC degradation by persulfate activation with carbon-coated CuFe2O4
Enhancement of anti-diabetic activity of pomelo peel by the fermentation of Aspergillus oryzae CGMCC23295-In vitro and in silico docking studies
Enhancement of metal-binding affinity for Cu+ Cu2+ complexes by hydrogen bond network
Enhancement of solubility and dissolution rate of dipyridamole by salifying-Preparation, characterization, and theoretical calculation
Enhancing operational stability of OLEDs based on subatomic modified thermally activated delayed fluorescence compounds
Enhancing photodynamic inactivation via tunning spatial constraint on photosensitizer
Enhancing Stability in Polynitrogen Compounds-Mechanisms and Strategies
Environment-friendly and high-performance azo dye substitutes for enhanced optical characteristics and color fastness
Equilibrium solubility, solvation and dissolution thermodynamics, and density functional theory study of albendazole in solutions of acetone-methanol-isopropanol + water
ESIPT and anti-Kasha behavior in hydroxy-aza-[n]cycloparaphenylenes
ETS -NOCV and IRI Analysis of the Amide Catalyzed Amide - Imidic Acid Tautomerism of Variously Substituted Amides
Excess Properties, Computational Chemistry, and Spectral Analysis of [Diethanolamine + Alcohols (Ethanol, n-Propanol, or n-Butanol)] Ion-Like Liquids
Excited dipole bound electronic states of potassium iodide anions-A theoretical perspective
Excited-state dynamics of C3-symmetric heptazine-based thermally activated delayed-fluorescence emitters
Exohedrally and endohedrally doped 15-crown-5 (a crown ether) surface by metals for potential applications as high-performance NLO materials
Experimental and Computational Studies on the Effects of C(2) Methylation on the Properties and Gas Separation Performance of Polyimide-Ionene Membranes
Experimental and DFT research for the effects of sodium on the heterogeneous reaction between NO and semichar derived from O2-CO2 pretreatments
Experimental and Theoretical Investigation on the Extractive Mass Transfer of Eu3+ Ions Using Novel Amide Ligands in 1-Hexyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)imide
Experimental and Theoretical Study of the New Leveler Basic Blue 1 during Copper Superconformal Growth
Experimental, Computational investigations and biological evaluation on 1-(3-Acetamidophenyl)-5-mercaptotetrazole- Cytotoxicity MTT assay
Explaining the selectivities and the mechanism of [3+2] cycloloaddition reaction between isoalantolactone and diazocyclopropane
Exploiting the (–C–H···C–) Interaction to Design Cage-Functionalized Organic Superbases and Hyperbases-A Computational Study
Exploration of charge transfer analysis and photovoltaics properties of A-D-A type non-fullerene phenazine based molecules to enhance the organic solar cell properties
Exploration of high-performance triptycene-based thermally activated delayed fluorescence materials via structural alteration of donor fragment
Exploration of nonlinear optical enhancement in acceptor–π–donor indacenodithiophene based derivatives via structural variations
Exploring Benzene Ru(II) Complexes of 2-Subsitituted Quinoline-Napthyridine Ligands-Synthesis, Biomacromolecular Binding and DFT Investigations
Exploring Differences in Lanthanide Excited State Reactivity Using a Simple Example-The Photophysics of La and Ce Thenoyltrifluoroacetone Complexes
Exploring novel naphthalene-fused octacyclic core-based non-fullerene acceptor materials with augmented optoelectronic attributes for stable and efficient solar cells
Exploring Robust Delayed Fluorescence Materials via Structural Rigidification for Realizing Organic Light-Emitting Diodes with High Efficiencies and Small Roll-Offs
Exploring the Dissolution Behavior of m-Hydroxybenzoic Acid in 14 Pure Solvents Using Thermodynamic, Molecular Simulation and Hansen Solubility Parameters
Exploring the effects of counterions and solvents on binuclear and tetranuclear Cu(II) oligo(N,O-donor) salamo-based complexes
Exploring the influence of the phosphorus-heteroatom substitution in Nnicotine on its electronic and vibrational spectroscopic properties
Exploring the promising application of Be12O12 nanocage for the abatement of paracetamol using DFT simulations
Exploring the Steric Effect in the Formation of Hydrogen Bonded Complexes of Isopropyl Amine with Aryl Ethers in n-Hexane
Exploring π-π Interactions and Electron Transport in Complexes Involving Hexacationic Host and PAH Guest
Exposing the Oxygen-Centered Radical Character of Tetraoxido Ruthenium(VIII) Cation
Extending the large molecule limit The role of Fermi resonance in developing a quantum functional group
Extractive Adsorption of Lactic Acid from Fermentation Broth on a Novel Ion Exchange Resin Impregnated by the [Bmim]PF6 Ionic Liquid
Facile Synthesis, Spectroscopic and Nonlinear Optical Insights of Hydrazinyl-Based Functional Materials
Facile Wet Chemical Synthesis of Dimeric Triangulene Derivatives through Intramolecular Radical-Radical Coupling
Fast and Selective Luminescent Sensing by Langmuir-Schaeffer Films Based on Controlled Assembly of Perylene Bisimide Modified with A Cyclometalated Au(III) Complex
FEATURES OF DICYANAMIDE BINDING TO A POLYNUCLEAR METALLAMACROCYCLIC COPPER COMPLEX
Five-membered vs. six-membered ESIPT in 3,5-dihydroxychromone
Flotation and adsorption of novel Gemini decyl-bishydroxamic acid on bastnaesite
Fluorescence property and solvent effect on m-bromosalicylaldehyde derivative; insights from synthesis, characterization, antimicrobial activity and computational studies
Fluorescent Origin and Chiral Nature of M?bius Carbon Nanobelts
Fluorination and Conjugation Engineering Synergistically Enhance the Optoelectronic Properties of Two-Dimensional Hybrid Organic–Inorganic Perovskites
Fluorination effects probed in 4-fluoroacetophenone and its monohydrate
Fluxional Hydrogen Bonds in Small Water Clusters (H2O)n (n?=?2–6)
Fractionation of poplar wood with different acid hydrotropes-Lignin dissolution behavior and mechanism evaluation
Free radical formation via BDE-209 thermolysis in the precalciner of a cement kiln-Simulation and DFT study
Free Radical-Mediated Photocyclization of Triphenylphosphindole Oxides for Photoactivated and Self-Reported Lipid Peroxidation
From Concept to Synthesis-Developing Heat-Resistant High Explosives through Automated High-Throughput Virtual Screening
From Helices to Crystals-Multiscale Representation of Chirality in Double-Helix Structures
Functionalization of fused imidazole-oxadiazole, triazole-oxadiazole and tetrazole-oxadiazole skeletons-Search for stable and potential energetic materials
Geometry optimization, impact of solvation on the spectral (FT-IR, UV, NMR) analysis, Quantum chemical parameters, and the bioactivity of feruloyltyramine as a potential anti-Lassa virus agent via molecular docking
Glyburide in a series co-solvent solutions-Solubility and modeling, solvation and quantum chemistry research
Graph Energy Variants and Topological Indices in Platinum Anticancer Drug Design-Mathematical Insights and Computational Analysis with DFT and QTAIM
Graphdiyne-porphyrin composite materials GDY-Por and Por@GDY for lithium ion battery anodes
Graphene Oxide-Based Aluminum Complex Ion Supercapacitor
Green and designable deep eutectic solvents for extraction separation of oxygenated compounds in Fischer-Tropsch oil products
Green synthesis and theoretical study of new 1,3,4-oxadiazoles-Application of Cu-Fe3O4@MWCNT magnetic nanocomposites
Grinding-induced supramolecular charge-transfer assemblies with switchable vapochromism toward haloalkane isomers
Halogen Bond-Aided Chiral Host–Guest Complexation between Iodoflurinated Benzene Derivatives and Molecular Tweezers
Halogen Bonding Involving Isomeric Isocyanide-Nitrile Groups
Harnessing the radical potential of calcium-activated biochar for non-peroxide catalytic degradation of bisphenol S
Heavy Pnicogen Atoms as Electron Donors in Sigma-Hole Bonds
Heptagon-Embedded Helicene Derivatives-Synthesis, Crystal Structural Analyses, and Circularly Polarized Luminescence
High pressure behaviour of the organic semiconductor salt (TTF-BTD)2I3
High-energy nitrogen rings stabilized by superatom properties
High-performance black polyimide with improved solubility for flexible printed circuit boards
Highlighting non-covalent interactions to molecular structure, electronic and vibrational spectra in a new hybrid organic-inorganic cobalt complex-synthesis, experimental and computational study
Highly efficient and thermally stable fullerene-free organic solar cells based on a rhodanine acceptor to enhance photovoltaic performance
Highly efficient color-tunable organic co-crystals unveiling polymorphism, isomerism, delayed fluorescence for optical waveguides and cell-imaging
Highly efficient construction of multi-substituted aminopyrazoles derivatives via iodine-mediated three-components reaction as potential anticancer ag
Highly efficient transition metal doped C24 electrocatalysts for hydrogen evolution reaction
Highly robust quantum mechanics and umbrella sampling studies on inclusion complexes of curcumin and β-cyclodextrin
Highly Solvent Resistant Small-Molecule Hole-Transporting Materials for Efficient Perovskite Quantum Dot Light-Emitting Diodes
Host–guest interactions in the solid state structure of a zinc(II) compound with a protonated diamine and DFT study
How polymer infiltration affects metal-organic frameworks-based facilitated hybrid membrane performances for CO2 separation
Humidity-Oxygen- Insensitive Organic Synaptic Transistors Based on Optical Radical Effect
Hybrid-ion strategy enables ultra-long life aqueous iron-organic batteries
Hydration Mechanism and Its Effect on the Solubility of Aripiprazole
Hydrazine Hydrate-Initiated GO-PAN Composite Fiber Cyclization Reaction to Achieve Chemical Stabilization of PAN
Hydrogen bond ferroelectric liquid crystal for tripartite temperature sensor-Experimental and computational (DFT) studies
Hydrogen-bonded organic framework-based bioorthogonal catalysis prevents drug metabolic inactivation
Hydrolysis and condensation of monobutyltin chloride-reaction process analysis with DFT
Hydroxyl-groups engineering of triazole-based polymers for efficient photocatalytic hydrogen peroxide production
Identification of interaction of 1,2,3,6-Tetrahydro-2,6-dioxo-4-pyrimidinecarboxylic acid using DFT Studies, molecular docking, biological activity
Identification of novel carbonic anhydrase II receptor-targeting drugs for treating myocardial infarction through the mechanism of Xue-Fu-Zhu-Yu decoction
Impact of ester-anchored alkyl side chains on the interfacial arrangement and charge transfer in organic solar cells
Impact of Polythiophene ((C4H4S)n; n = 3, 5, 7, 9) Units on the Adsorption, Reactivity, and Photodegradation Mechanism of Tetracycline by Ti-Doped Graphene
Impact of π-Linkers Modifications on Tuning Nonlinear Optical Amplitudes of Pyridoquinazolinone-Based Aromatic Dyes
Implementation of machine learning protocols to predict the hydrolysis reaction properties of organophosphorus substrates using descriptors of electron density topology
Importance of solvent roles in molecular, electronic and dynamical properties, thermodynamic quantities, Mulliken charges, reactive analysis and molecular docking of 2-Bromo-1H-imidazole-4,5-dicarbonitrile
Importance of solvents role in molecular and electronic properties, DFT, spectroscopic, Electron-hole transition, chemical reactivity, topology and molecular docking investigations
Improved electrochemical detection of copper ions in nitrogen doped ta-C films for marine corrosive monitoring
Improved Prediction of Reaction Kinetics for Amine Absorbent-based Carbon Capture using Reactive Site-based Transition State Conformer Search Method
Improving predictions and understanding of primary and ultimate biodegradation rates with machine learning models
In silico approaches for the identification of novel ULK1 inhibitors-pharmacophore model, molecular docking and molecular dynamics simulations
In situ ligand-modulated activation of inert Ce(III-IV) into ozonation catalyst for efficient water treatment
In vitro cytotoxicity evaluation of organotin(IV) complexes derived from bisphosphoramide ligand
In-Silico aided screening and characterization results in stability enhanced Novel Roxadustat co-crystal
In-silico investigation of 4-nitro-N-1H-pyrazol-3-ylbenzamide towards its potential use against SARS-CoV-2
In-situ synthesis of dinuclear Iron (III) complex; Crystal structure, DFT calculations, Hirshfield surface analysis, and molecular docking.
Inclusion Complexes of Metastable-State Photoacid with High Acidity and Chemical Stability
Influence of azacycle donor moieties on the photovoltaic properties of benzo[c][1,2,5]thiadiazole based organic systems-a DFT study
Influence of nitroamino?nitroimino tautomerism-A useful theoretical supplement for nitroamino-based energetic materials
Influence of the solvents through molecular structure, wavefunction studies, biological activity prediction and molecular docking studies
Influence on the volatilization of ethyl esters-Nonnegligible role of long-chain fatty acids on Baijiu flavor via intermolecular interaction
Insertion of molecular hydrogen into slit-shaped carbon pores-theoretical study
Insight into (Z)-Ethyl-2-(2-((E)-2,4-dinitro benzylidene amino)-4-oxo-3-phenylthiazolidin-5-ylidene) acetate in-silico anti-SARS-CoV-2 performance
Insight into a novel cobalt complex with promising electric energy stocker properties-A combined DFT and experimental study
Insight into functional groups of salt compounds on the performance of viscoelastic surfactant-based fluids
Insight into the effect of side chains on thermal transport of organic semiconductors
Insight into the evolution upon ionization from tin-oxo cage photoresist and counterions by DFT calculation
Insight into the interaction of pluronic F-127 functionalized boron nitride nanosheets with piezoelectric poly(l-lactic acid) nanofiber scaffolds for bone tissue engineering applications
Insight into the selective oxidation behavior of organic pollutants via Ni-N4-C mediated electron transfer pathway
Insight into the shear stability of the polymer-metal complexes-Experiment and density functional theory studies
Insight into Understanding the Inverse Isotope Effect in a One-Dimensional Spin-Peierls-Type Molecular Solid
Insight of interaction in kojic acid-urea derivatives deep eutectic solvent from physicochemical characterization and computation simulation
Insight on the structural, electronic and optical properties of Zn, Ga-doped-dual-doped graphitic carbon nitride for visible-light applications
Insights into ball milling treatment promotes the formation of starch-lipid complexes and the relation between multi-scale structure and in vitro digestibility based on intermolecular interactions
Insights into the mechanism of electric field regulating hydrogen adsorption on Li-functionalized N-doped defective graphene
Insights into the mechanism of [3+2] cycloaddition reactions between N-benzyl fluoro nitrone and maleimides, its selectivity and solvent effects
Insights into the Zintl Cluster Mediated Disulfide Bond Separation
Insights into Unconventional Intramolecular CH???CO Interaction in Os3(CO)9(μ-H)2(μ3-η1- η1- η2-C16H8) Crystal
Insights into vibrational and electronic properties of (6,6)-phenyl-C61-butyric acid methyl ester (PCBM) chemical bonding with (CuO)n clusters
Insights of the pH-dependent emission of carbon dots a density functional theory study
Interaction behavior study of HCl on (ZnS)n (n?=?1–12) clusters and HCl effect on Hg0 adsorption
Interaction of serotonin and histamine with water and ethanol-Evidence from theoretical investigations
Interactions of adenine–thymine base pair with potassium cation and water clusters
Intermolecular charge-transfer complex between solute and ionic liquid-experimental and theoretical studies
Introducing the various electron withdrawing functions in trigging the optical nonlinearity of benzodithiophene based chromophores
Investigating sulfonamides - Human serum albumin interactions-A comprehensive approach using multi-spectroscopy, DFT calculations, and molecular docking
Investigating the Chemical Reactivity of Kahalalides-A Promising Source of Therapeutic Peptides from Marine Natural Products
Investigating the influence of substituent groups in TTM based radicals to the excitation process
Investigating two structurally different penta-coordinated trigonal bipyramidal Co(II) non-symmetric salamo-type complexes
Investigation into Pigmentation Behaviors and Mechanism of Pigment Yellow 180 in Different Solvents
Investigation of Molecular Structure, Topological and Molecular Docking Studies of a Novel Anticancer Drug Pacritinib
Investigation of structural, spectral, and electronic properties of complexes resulting from the interaction of acetonitrile and hypohalous acids
Investigation of the Effect of Solvents on the Synthesis of Aza-flavanone from Aminochalcone Facilitated by Halogen Bonding
Investigation of the synergistic effect of O2-H2O pretreatment and Na addition on the NO-semichar reaction
Investigation of typical heterocyclic fragment structures and reaction characteristics in oil shale using density functional theory
Investigation of γ-irradiation dependent structural properties of LLM-105?by ultraviolet-visible diffuse reflectance spectroscopy
Investigation on solid-liquid equilibrium behavior of 4-cyanobenzoic acid in fourteen mono-solvents
Investigation on the stability of the Enol Tautomer of Favipiravir and its derivatives by DFT QTAIM NBO NLO and 1H-NMR
Ion-exchange purification, Nano-HPLC–MS MS identification and molecular dynamics simulation of novel umami peptides from fermented grain wine
Is there a Covalent Au(I)–Au(I) Bond in the trans-(AuX)2 (X = F, Cl, Br, I) Structure?
Isomeric Thiadiazole-xLi+(x = 1, 2) templates found to be efficient in search of potential H2 storage systems; a computational study
Isosterism in pyrrole via azaboroles substitution, a theoretical investigation for electronic structural, stability and aromaticity
Kokumi-Enhancing Mechanism of N-l-lactoyl-l-Met Elucidated by Sensory Experiments and Molecular Simulations
Large-molecular-weight PVP-functionalized stacked upconversion nanoplates for high-performance individual marking
Lattice-Selective Cross-Interface Bidentate Passivation for Efficient Bifacial Tandem Carbon-based Perovskite Solar Cells
Ligand and Substituent Effect on Regium?π Bonding in Cu and Ag π-Conjugated Complexes-A Density Functional Study
Ligand Effects on Isomerization-Bond-Formation Networks in Stereodivergent Pd-Cu Dual Catalysis Involving Stereochemically Dynamic Allylpalladium Intermediates
Linear and Nonlinear Optical Responses of Nitrobenzofurazan- Sulfide Derivatives DFT-QTAIM Investigation on Twisted Intramolecular Charge Transfer
Linear and Nonlinear Optical Responses of Nitrobenzofurazan-Sulfide Derivatives
Lithium isotope Separation by precipitation method coupled with solvent extraction with 1-Hydroxy-4-(p-toluidino) anthraquinone
Locking water molecules via ternary O-H···O intramolecular hydrogen bonds in perhydroxylated closo-dodecaborate
Lone-pair, Electron-dominated, Nonlinear Optical Responses in Sulfur Clusters and Electric Tunability Properties
Low-cost Fully Non-Fused Ring Acceptor Enables Efficient Organic Photovoltaic Modules for Multi-scene Applications
Low-temperature structural deformation and fragmentation of lead styphnate by in-situ experiments and calculation
Low-Temperature-Resistant Elastic Organic Crystals with Variable Amplified Spontaneous Emissions
Luminescent four-coordinate N-heterocyclic carbene (NHC) copper(I) complexes bearing the pyridyl-imidazolylidene ligand
Luminescent Heterobimetallic Au(I)-Ag(I) Complex Stabilized by a Unique Acetylide Fragment
Machine learning methods for developments of binding kinetic models in predicting protein-ligand dissociation rate constants
Machine learning prediction of delignification and lignin structure regulation of deep eutectic solvents pretreatment processes
Macrophage-Inspired Degradation-Activation System Enables Ultrasensitive Multicolor Detection of Haloacetic Acids via Janus Perovskite Halide Exchange
Manipulating fluorine induced bulky dipoles and their strong interaction to achieve high efficiency electric energy storage performance in polymer dielectrics
Manipulating nitration and stabilization to achieve high energy
Manipulating the ESPT process and photophysical properties of HQCT by water-based hydrogen bond bridge
Markovnikov vs anti-Markovnikov Iodine-Mediated Acyloxyselenenylation of Alkenes and Dienes with Elemental Selenium and Carboxylic Acids
Measurement of Solubility and Molecular Simulation of Dissolution Behavior of Isonicotinamide in 15 Pure Solvents
Measurement, Correlation, and Analysis of the Solubility of Triethylamine Hydrochloride in ten Pure Solvents
Mechanism Analysis and Property Prediction of Extended Surfactants Based on the Respectively Optimized Force Field
Mechanism and dynamics of Baeyer–Villiger oxidation of furfural to maleic anhydride in presence of H2O2 and Au clusters
Mechanism Insight into the Conversion between H2S and Thiophene during Coal Pyrolysis
Mechanism of disproportionation of methylchlorosilanes by ZSM-5(3T)@MIL-53(Al) core–shell catalyst
Mechanism of photocatalyzed imine conversion-a relayed process of sequential energy transfer, single electron transfer and proton transfer
Mechanisms of Cu(II) enhancing photolysis of tetracycline under UV irradiation
Mechanisms of ssDNA aptamer binding to Cd2+ in aqueous solution-A molecular dynamics study
Mechanistic Details of the Titanium-Mediated Polycondensation Reaction of Polyesters A DFT Study
Mechanistic insights into photoinduced energy and charge transfer dynamics between magnesium-centered tetrapyrroles and carbon nanotubes
Mechanistic Investigation of Isobutanol-Isobutyl Acetate Separation by Extraction Using Low-Transition Temperature Mixtures
Mechanistic Study on the Formation of the Alkyl Acrylates from CO2, Ethylene and Alkyl Iodides over Nickel-based Catalyst
Metal charge-directed enantiodivergent asymmetric transfer hydrogenation of ketones
Metal-doped fullerenes as promising drug carriers of hydroxycarbamide anticancer-insights from density functional theory
Metal-Metal Bonds with Unusual Oxidation States in Early s-Block Elements-A Computational Perspective
Metallophilic and Inter-Ligand Interactions in Diargentous Compounds Bound by a Geometrically Flexible Macrocycle
Methoxylated Quinoline-Chalcones with Potential Pesticidal Activity From Synthesis to Supramolecular Framework
Microfluidic Printing of Vertically-Oriented Nanosheets-MOFs Hetero-Interface for Intensive Pseudocapacitive Storage
Microwave-assisted solvent-free synthesis, Hirshfeld surface analysis and DFT studies of 1-(hydroxymethyl)-pyrazoles
Migration mechanism of atrazine in the simulated lake icing process at different freezing temperatures based on density function theory
Mitochondria-targeted ruthenium(II)-based phosphorescent chemodosimeter for peroxynitrite detection in drug-induced liver injury
Mobile hydrogen-bonding donor in covalent organic framework for efficient iodine capture
Modeling of DNA interactions with anti-cancer activity drugs using the ONIOM method
Modified superhydrophobic magnetic Fe3O4 nanoparticles for removal of microplastics in liquid foods
Modifying the electron capture decay rate of 7Be by using small fullerenes
Modulating organic photovoltaic properties of non-fullerene acceptors by molecular modification based on Y6
Molecular Adsorption-Induced Interfacial Solvation Regulation to Stabilize Graphite Anode in Ethylene Carbonate-Free Electrolytes
Molecular aggregation kinetics of Heteropolyene-An Experimental, topological and solvation dynamics studies
Molecular design and experimental study of the conversion of cellulose to 5-HMF catalyzed by different ratios of Br?nsted -Lewis acidic deep eutectic solvents
Molecular Dynamics Simulation of CO2 Hydrate Growth in NaCl Aqueous Solution.png
Molecular dynamics simulation of potassium perfluorooctanesulfonate at the oil-water interface
Molecular dynamics simulation of the distribution of potassium perfluorooctanesulfonate in water
Molecular Dynamics Study of Silica Nanoparticles and CO2-Switchable Surfactants at an Oil–Water Interface
Molecular dynamics study on the dissolution characteristics and cluster formation of Li2CO3 in supercritical water
Molecular identification of organic acid molecules from α-pinene ozonolysis
Molecular Insights into Cucurbit[8]uril?Mediated Complexes-Enhanced Interaction Cooperation towards Pseudostatic Dynamics - 副本
Molecular Insights into Cucurbit[8]uril?Mediated Complexes-Enhanced Interaction Cooperation towards Pseudostatic Dynamics
Molecular ionization potentials of triglycerides and mechanism analysis of streamer discharge in vegetable insulating oil
Molecular modelling of Al24N24 nanocage for the chemical sensing of phosgene and mustard chemical warfare agents-First theoretical framework
Molecular orbital and topological electron density study of n → πx interactions-amides and thioamides cases
Molecular scale behavior of xylan during solvent-controlled extraction and precipitation
Molecular simulation and experimental insights into the separation of dimethyl carbonate and isopropyl alcohol using imidazolium ionic liquids
Molecular structure, physicochemical properties, Impact of solvents ionization potential, electron occupancy, inhibition constant, and stabilization energy investigations of 4-Acetamido benzoic acid
Monoterpenoid indole alkaloid dimers from the Melodinus axillaris induce G2-M phase arrest and apoptosis via p38 MAPK activation in HCT116 cells
Multi-resonance thermally activated delayed fluorescence molecules with intramolecular-lock-theoretical design and performance prediction
Multifunctional Dapsone Additives Prepare Efficient and Stable Perovskite Solar Cells
Multinuclear Pd(II) oligomeric nanohoop complexes with 2,2′-bipyridyl-embedded cycloparaphenylenes ligands exhibiting ultrahigh third-order nonlinear optical performance
N-Indolyl diiron vinyliminium complexes exhibit antiproliferative effects in cancer cells associated with disruption of mitochondrial homeostasis, ROS scavenging, and antioxidant activity
Nano-assembled modified graphene composites based on rapid microwave fabrication for thermal management
Nano-enhanced drug delivery of dacarbazine using heteroatoms (B, N, P, S) doped Ag-functionalized silicene nanomaterials
Natural deep eutectic solvents of γ-valerolactone and formic acid for oxidative desulfurization with superior efficiency
Nature of a Tetrabutylammonium Chloride–Levulinic Acid Deep Eutectic Solvent
Nature of Chemical Bond in BeF? Anion-A Charge Shift Bond From Electron Density Signatures
Nature-inspired construction of multifunctional composited membrane by in situ formation TA-APTES NPs for selective adsorption of tetrabromobisphenol a and oil-water separation
New insights into the structure-activity relationship for CO2 capture by tertiary amines from the experimental and quantum chemical calculation perspectives
Nitrogen Atom Induced Contrast Effect on the Mechanofluorochromic Characteristics of Anthracene-Based Acceptor-Donor-Acceptor Fluorescent Molecules
Noncovalent Interactions of Aromatic Heterocycle-Rotational spectroscopy and theoretical calculations of the thiazole-CF4 and thiazole-SF6 complexes
Novel insights into the effects of asphaltenes on oil phase properties
Novel jellyfish-shaped resorcin[4]arene macrocyclic cocrystal via collaboration of macrocyclic chemistry and co-crystal engineering
N?N bridged pyrazoles from nitrogen-centered radicals-A comparative study of spin densities, molecular structures, and reactivities
Occurrence and Formation Mechanism of PCDD-Fs and SCCPs in Chlorinated Paraffin Products
On the Aromaticity of Puckered Ions C4H42+ and B4H42? Deciphering the Origin of Nonplanarity
On the importance of unconventional Cu?π interaction in tetrachloro-bis(1,10-phenanthroline)-dicopper(II) complex-Insights from experiment and theory
On-surface synthesis of nitrogen-doped nanographene with an [18]annulene pore on Ag(111)
One-dimensional molecular co-crystal alloys capable of full-color emission for low-loss optical waveguide and optical logic gate
One-Step Synthesis of Fragment-Reduced Graphene Oxide as an Electrode Material for Supercapacitors
One-step synthesis of Janus hydrogel via heterogeneous distribution of sodium α-linoleate driven by surfactant self-aggregation
Open-Cage Fullerene as a Macrocyclic Ligand for Na, Pt, and Rh Metal Complexes
Optical Spectrum of Tetrafluorosubstituted Zinc Phthalocyanine
Optimal preparation of pyrene magnetic molecularly imprinted polymers by simulation calculation and study of its adsorption properties
Optimizing water dissociation dehydrogenation process via Sn single atom incorporation for boosting photocatalytic CO2 methanation
Organosulfur and Organoselenium Functionalized Benzimidazo[1,2-a]quinolines
Oriented External Electric Field Modulated Superhalogen Characteristic and Photoelectric Properties of Al4N2 Cluster
Outstanding inhibition of H2O2 generation in doubly doped graphene-The synergy of two heteroatoms opens a new chemical path
Overlooked Solid State Structure of 1,3-I2C6F4—The Meta-Member of an Iconic Halogen Bond Donors Trio
Oxidative Addition and β-hydride Elimination by a Macrocyclic Dinickel Complex Observing Bimetallic Elementary Reactions
Pd and Pt Metal Atoms as Electron Donors in σ-Hole Bonded Complexes
Perception of Insight in the Formation of Solid Electrolyte Interphase
Performance evaluation and efficiency of functionalized cellulose nanocomposites for the removal of pharmaceutical contaminants from the aqueous environment
Peripheral structural modification for devising push–pull strategy into 1,3,5-triaryl-2-pyrazoline-based compounds for nonlinear optical insights via density functional theory approach
Phase equilibria and mechanism analysis of separating ethanol from fuel additives by chord chloride-deep electrochemical solvents
Photocatalytic 1,2-Phosphorus-Migrative [3 + 2] Cycloaddition of Tri(t-butyl)phosphine with Terminal Alkynes
Photocatalytic degradation of different antibiotics by TiO2-carbon composites-a case study of tetracycline and ciprofloxacin
Photoluminescent and Magnetic Properties of Mononuclear Lanthanide-Based Compounds Containing the Zwitterionic Form of 4-Picolinic Acid as a Ligand
Photostimulated Covalent Linkage Transformation Isomerizing Covalent Organic Frameworks for Improved Photocatalytic Performances
Photovoltaic and Optical Characterization of Organic Heterocyclic 6,6′-Dibromoindigotin Crystal Under Changing Solvent Polarity by DFT Analysis
Physicochemical Characterization of Clay and Study of Cationic Methylene Blue Dye Adsorption
Piezo-photocatalysis of Sr-doped Bi4O5Br2-Bi2MoO6 composite nanofibers to simultaneously remove inorganic and organic contaminants
Piezoelectric charges elicited a chain reaction of sulfite activation-Performance and mechanism
Polyanion-induced single zinc-ion gel polymer electrolytes for wide-temperature and interfacial stable zinc-ion hybrid capacitors
Polymorphism and Multi-Component Crystal Formation of GABA and Gabapentin
Predictive models for metal-metal bond dissociation free energies between aluminum(III) and a series of transition metal carbonyls
Preparation of a Compound With Si(II)–Si(IV)–Si(II) Bonding Arrangement
Pristine and vacancy defective boron nitride nanotubes absorb deep eutectic solvents
Probing Near-infrared Absorbance of E and Z Diazene Isomers via Antiaromaticity
Probing the catalytic activity of first-row transition metal doped C20 fullerene as remarkable HER electrocatalysts-A DFT study
Probing the effect of magnesium doping on the structural and electronic properties of boron clusters
Probing the REDOX effect of helical tetraspirobenzene on nonlinear optical properties
Promising bioactive properties of (2R,5S)-2,5-dimethylpiperazine-1,4-diium dinitrate material-experimental, theoretical and in silico investigation
Promising impact of push–pull configuration into designed octacyclic naphthalene-based organic scaffolds for nonlinear optical amplitudes
Promoting through-space charge transfer-based TADF via rational alignment of quasi-planar donor and acceptor in solid state
Protein3D-Enabling analysis and extraction of metal-containing sites from the Protein Data Bank with molSimplify
Psoralidin–cucurbit[7]uril complex with improved solubility to tackle human colorectal cancer-experimental and computational study
Push-Pull and Conventional Nitriles as Halogen Bond Acceptors in their Cocrystals with Iodo-substituted Perfluorobenzenes
QSPR models for sublimation enthalpy of energetic compounds
Quantifying the Electronic Effect of C4 and C4' Positions of Bipyridine Ligand on Pd(II)-Rh(I)-Catalyzed Conjugate Addition Reaction
Quantum calculation assisted efficient lithium extraction from unconventional oil and gas field brine by β-diketone synergic system
Quantum Chemical Insight into 1,2-Shift Rearrangement in Bromination of Allylaryls
Quantum chemical study on the catalytic debromination mechanism of brominated epoxy resins
Quantum mechanical assessment on the optical properties of capsanthin conformers
Quasi-open Cu(i) sites for efficient CO separation with high O2-H2O tolerance
Raman spectroscopy and quantum chemical calculation on YCl3-KCl molten salt system
Rare formyl-coordinated homotrinuclear copper(II) salamo-based N2O3-donor complex-Experimental and theoretical studies
Rational design of AIEE sensor-Ultrafast detection of methanol in liquid and gaseous states with highly sensitive and selective performance
Rational design of co-ordination compounds in combination of bipyridine type of ligands and group 7 metal (M = Mn, Re) for photoCORM
Rational design of soluble intrinsic black polyimide containing tetraphenylcyclopentadienone-based chromophore
Re-establishing the Lost Covalency of the Inverted Bond in [1.1.1]Propellane
Reaction Mechanism and Effect of Substituent in Direct Bromination of Indoles
Reaction Mechanism and Kinetics for the Selective Hydrogenation of Carbon Dioxide to Formic Acid and Methanol over the [Cu2]0,±1 Dimer
Reaction mechanism of sodium with hydrogen fluoride in the formation of sodium fluoride and the adsorption of hydrogen fluoride on sodium fluoride monomer and tetramer
Reaction mechanism of ZrAln (n=1-4) clusters and water molecules-A density functional theory study
Reactivity and Structural Investigation of Tetrahydroneoprzewaquinone A as an Anti-Inflammatory Agent
Realizing long range π-conjugation in phenanthrene and phenanthrene-based molecular crystals for anomalous piezoluminescence
Red thermally activated delayed fluorescence emitters based on phenazine-derived electron donors and solution-processed OLEDs
Redox-Activated NO Release in Monolayer Regime
Regioselective Direct C–H Bond Heteroarylation of Thiazoles Enabled by an Iminopyridine-Based α-Diimine Nickel(II) Complex Evaluated by DFT Studies
Regioselective Synthesis of 2,4- and 2,5-disubstituted 1,3-thiazoles from 2-oxo-2-(amino)ethanedithioates via Base Catalysed Cyclization
Regulating fluorescent properties and ESIPT behavior of novel flavone-based fluorophore by replacing oxygen atom in proton donor-acceptor with sulfur atom
Reliable and accurate prediction of basic pK_a values in nitrogen compounds-the pK_a shift in supramolecular systems as a case study
Remote stereocontrol with azaarenes via enzymatic hydrogen atom transfer
Research on solubility behavior of iminostilbene in twelve mono-solvents-Measurement, modeling, molecular simulation and thermodynamic properties
Research on the reaction path of chlorobenzene oxidation by electrochemical-sodium persulfate system
Restricted intramolecular vibrations assisted enhanced fluorescence emission response of probe
Revealing the contributions of DFT to the spectral interpretation for an amino benzoyl thiourea derivative
Revealing the Mechanism of Glutamic Acid-Functionalized Layered Double Hydroxides and its Removal Mechanism for Cr(VI) from Microscopic and Macroscopic Perspectives
Reversible and irreversible stimuli-responsive chromism of a square-planar platinum(II) salt
Role of excited-state hydrogen bonding in CO2 photoreduction catalyzed by sodium magnesium chlorophyll
Role of extended end-capped acceptors in non-fullerene based compounds towards photovoltaic properties
Role of Maillard reactions in co-pyrolysis of lignite and spent coffee grounds-Fixation of nitrogen in semicoke and improved adsorption of phenols
Role of solvents in molecular level interaction, reactivity and spectral characterisation of 2-Amino-3-(((E)-4-(dimethylamino)benzylidene)amino) maleonitrile-Anti depressant agent
Role of the solvent polarity on the optical and electronic characteristics of 1-iodoadamantane
Role of π-Radical Localization on Thermally Stable Cross-Links Between Polycyclic Aromatic Hydrocarbons
Room-temperature synthesis of magnetic thiophene-based covalent organic frameworks for derivatization-assisted GC–MS analysis of estrogens in environmental water
Route to Stabilize Cubic Gauche Polynitrogen to Ambient Conditions via Surface Saturation by Hydrogen-1
Salt-Induced Coil–Globule Transition of Sulfonate-Modified HPAM is Affected by the Branched Chain Length
Scavenging of “Dead Sulfur” and “Dead Lithium” Revealed by Integrated–Heterogeneous Catalysis for Advanced Lithium–Sulfur Batteries
Screening for the adsorption-activated H2O2 and peroxymonosulfate for high-performance heteroatom-doped graphene
Selective adsorption of 1-hydroxyethylidene-1,1-diphosphonic acid on calcite surface in smithsonite flotation system
Selective Confinement of Potassium, Rubidium, or Caesium Ions in a Non-covalent Hydroxyproline Octamer Cage Stabilized by Cis-hydroxyl Locks
Self-acclimation mechanism of pyrite to sulfamethoxazole concentration in terms of degradation behavior and toxicity effects caused by reactive oxygen species
Self-Assembled Lenalidomide-AIE Prodrug Nanobomb for Tumor Imaging and Cancer Therapy
Self-gelation techniques for amoxicillin-formulation and characterization
Self-Reporting Photodynamic Nanobody Conjugate for Precise and Sustainable Large-Volume Tumor Treatment
Separation of fuel additives based on mechanism analysis and thermodynamic phase behavior
Significant influence of water molecule on the SO3 + HCl reaction in the gas phase and at the air-water interface
Similar but Not Same-Impact of Structurally Similar Coformers on Co-crystallization with Telmisartan
Simulation of exciton spectra in disordered supramolecular polymers-Exciton localization in cisoid indolenine squaraine hexamers
Simultaneous identification of strong and weak interactions with Pauli energy, Pauli potential, Pauli force, and Pauli charge featured
Simultaneous leaching and separating of palladium from spent palladium catalyst by tribromide ionic liquids as non-volatile oxidizing solvents
Simultaneously regulating absorption capacities and antioxidant activities of four stilbene derivatives utilizing substitution effect
Single Vacancy-Defected Endohedral Metallofullerene-Superhalogen
Single-atoms (N, P, S) encapsulation of Ni-doped graphene-PEDOT hybrid materials as sensors for H2S gas applications
Single-crystal structure and theoretical calculations of the second ternary tellurium borate Te2B2O7
Size-tunable energy gaps of hydrogen-terminated biphenylene segments
Solid State Phosphorescence Enhancement of PtII-Based Emitters via Combination of π-Hole(Isocyano Group)??? dz2[PtII] and I···Cl Halogen-Bonding Interactions
Solubility determination, dissolution properties and solid transformation of resmetirom (form A) in heptane and seven alcohols
Solvation effect on photophysical properties and ESIPT behaviours for 2-benzooxazol-2-yl-6-thiophen-2-yl-phenol fluorophore
Solvation Impact on the Geometry and Electrical Properties of Flufenoxuron-A Topological and Spectral Insight by DFT Approach
Solvent impact on electronic, photochemical, molecular structure, topology studies, and the antihistamine activity of 2-(2-Benzylphenoxy)-N,N-dimethylethanamine
Solvent role in electronic energies, Experimental Vibrational studies, Surface and biological analysis of Ethyl Gallate
Solvent-regulated fluorimetric differentiation of Al3+ and Zn2+ using a dual functional sensor based on the photo-induced electron transfer and intramolecular proton transfer mechanism
Solvent-solute interaction, Thermodynamic behaviour, structural, chemical and anti-cancer biological properties of 3(2h)-furanone derivatives
Solvents performance, experimental spectral energies interactions, excitations energies, topological and electrophilic molecular interaction studies
Spectroscopic (FT-IR, FT-Raman, UV-Vis and NMR) and computational (DFT, MESP, NBO, NCI, LOL, ELF, RDG and QTAIM) profiling of 5-chloro-2-hydroxy-3-methoxybenzaldehyde
Spectroscopic and computational studies of the noncovalent interactions in the structures of the 2-hydroxy-3-iodo-6-methoxybenzenecarbonyl (-C(O)CH3, -CHO) derivatives
Spectroscopic investigations and electronic transitions, topology studies, and biological assay of a potent antimicrobial compound
Spectroscopic, Biological, and Topological Insights on Lemonol as a Potential Anticancer Agent
Spiro Boron-Nitrogen Molecules Based Thermally Activated Delayed Fluorescence Emitter for Highly Efficient Solution-Processed Organic Light-Emitting Diodes
Spodium Bonds Involving Methylmercury and Ethylmercury in Proteins-Insights from X-ray Analysis and Computations
Stability and electronic structure of the magnetic hyperhalogen Fe(BF4)4
Stability Trend Analysis of Light Lanthanide Complexes with a Fluorophenyl-dipicolinamide-A Quantum Chemical Study
Stability, Electronic and Optical Properties of Irida-Naphthalene and Irida-Azulene-A Computational Investigation
Stabilization of Cyclic C4 by Four Donor Ligands-A Theoretical Study of (L)4C4 (L = Carbene)
Strained thiacyclophanes-Reducing properties and gauge of transannular interactions
Stress-Stabilized Crystalline Phases of Ultrahigh Molecular Weight Polyethylene under Tensile Stress
Strong carbon – noble gas covalent bond and fluxionality in hypercoordinate compounds
Structural Analysis and Reactivity Insights of (E)-Bromo-4-((4-((1-(4-chlorophenyl)ethylidene)amino)-5-phenyl-4H-1,2,4-triazol-3-yl)thio)-5-((2-isopropylcyclohexyl)oxy) Furan-2(5H)-one
Structural analysis, reactivity descriptors (HOMO-LUMO, ELF, NBO), effect of polar (DMSO, EtOH, H2O) solvation, and libido-enhancing potential of resveratrol by molecular docking
Structural analysis, reactivity descriptors (HOMO-LUMO, ELF, NBO), effect of polar (DMSO, EtOH, H2O) solvation, and libido-enhancing potential
Structural Design, and Biological Assessment of Co2+, Ni2+, Cu2+and Zn2+ complexes with N-(phenyl)-2-imino-2H-chromene-3-carboxamide
Structural deterioration of calcium aluminosilicate hydrate by sulfate attack
Structural elucidation and in-silico evaluation of 1,2,4-triazole derivative as potent Omicron variant of SARS-CoV-2 spike protein inhibitor with pharmacokinetics ADMET and drug-likeness predictions
Structural evolution and electronic properties of medium-sized CrSin? 0 (n = 19–25) clusters
Structural Evolution and Electronic Properties of Two Sulfur Atom-Doped Boron Clusters
Structural evolution and stability of plutonium oxide clusters
Structural evolution of carbon foam and its effect on polypyrrole-carbon foam composite electrodes in supercapacitors
Structural evolution, charge transfer and bonding properties of medium-sized atomic rubidium-doped boron clusters
Structural evolution, stability, electronic and bonding properties of sodium-doped magnesium cluster cations
Structural Isomers-Small Change with Big Difference in Anion Storage
Structural modeling of 1,4 azaborine-based chromophores for organic solar cells using bracing units with benzothiophene-incorporated acceptors for exploration of photovoltaic behavior
Structural, chemical reactivity, topological, electronic and spectroscopic (Raman, SERS and UV-Vis) and antiviral investigation of Furo [3,2-c] pyridine-2-carbaldehyde
Structural, electronic, intermolecular interaction, reactivity, vibrational spectroscopy, charge transfer, Hirshfeld surface analysis, pharmacological and hydropathy plot on 5-Bromo nicotinic acid
Structural, Energetic and Dynamic Investigation of Poly(ethylene oxide) in Imidazolium-Based Ionic Liquids with Different Cationic Structures
Structure-directing role of CH?X (X = C, N, S, Cl) interactions in three ionic cobalt complexes-X-ray investigation and DFT study using QTAIM Vr predictor to eliminate the effect of pure Coulombic forces
Study of Intramolecular Interactions in the Polyoxovanadate Cluster [(SO4) ? V16O42]6–
Study of the electronic effect and quantitative spectra predictions of o-methoxyaniline-terminated monoazonaphthols
Study on benzylidine derivatives as corrosion inhibitors for mild steel in 15% HCl medium
Study on curing kinetics of epoxy system and DFT simulation
Study on synthesis, surface activity and quantum chemical properties of anionic-nonionic Gemini surfactant
Study on the Mechanism of Ru-Catalyzed Cyclization of Aromatic Amides with Allylphosphine Oxides
Substituent effect on the intramolecular hydrogen bond and the proton transfer process in pyrimidine azo dye
Substituent-Induced Hyperconjugation-Origin of the Structural Effects on the Efficiency of Photochemical Ring Opening
Successive Protonation of Decaniobate, [Nb10O28]6? Electronic Properties and Spectra
Sulfite activation by Jahn-Teller-driven oxygen vacancies Cu-Mn composite oxide for chlortetracycline degradation
Sulfur Centered Hydrogen Bonding in Thioglycolic Acid and Its Clusters-A Computational Exploration
Supramolecular assembly involving weak hydrogen bonds, anti-parallel π···π stacking and O···C tetrel bonding interactions and LOX studies
Surface engineering of non-platinum-based electrocatalysts for sustainable hydrogen production
Surface enhanced Raman scattering investigation of tecovirimat on silver, gold and platinum loaded silica nanocomposites
Sustainable and effective gold(I) separation from aurocyanides wastewater and its mechanism using guanidinium ionic liquids
Synergetic Inhibition and Corrosion-Diagnosing Nanofiber Networks for Self-Healing Protective Coatings
Synergistic Advancement of Molecular Design and Dual Encapsulation Technology for High-Performance Room-Temperature Barocaloric Refrigeration Materials
Synergy of Copper Vacancies and Amorphous Regions in Copper Sulfides Enables Superior Capacity for Hg0 Adsorption
Synthesis and characterization of a new chalcone-based nonlinear optical crystal-BBC
Synthesis and Characterization of U≡C Triple Bonds in Fullerene Compounds
Synthesis and theoretical investigation of phenanthrodithiophene diimide
Synthesis of a new μ-chlorido-bridged tetra-nuclear copper(II) complex containing 2,3,5,6-tetra(2-pyridyl)pyrazine (tppz), chlorido and perchlorato ligands
Synthesis of bisindolyl diphenylene from its ketone derivatives by infrared irradiation supported on a natural clay
Synthesis of Polycyclic Aromatic Hydrocarbons with Highly Twisted N-Doped Heptalene
Synthesis, Antifungal Activity, Molecular Docking Studies, RDG Analysis, and DFT Computations on Structural Vibrational and Electronic Spectra of 3,5-Diamino-1,2,4-Triazolinium Picrate
Synthesis, characterization, biological evaluation, DFT and molecular docking studies of (Z)-2-((2-bromo-4-chlorophenyl)imino)methyl)-4-chlorophenol and its Co(II), Ni(II), Cu(II), and Zn(II) complexes
Synthesis, Characterization, DFT Studies and Biological Assessment of Palladium(II) Complexes with Imidazole Derivatives
Synthesis, Characterization, DFT, Vibrational analysis (FT-IR and FT-Raman), Topology and Molecular docking studies of 3,3′-((1E,1′E)-((sulfonylbis(4,1-phenylene)) bis (azaneylylidene)) bis (methaneylylidene)) diphenol
Synthesis, chemical bonding and reactivity of new medium-sized polyarsenides
Synthesis, crystal structure and hydrogen bonding of a Cp Rh(III) complex bearing pyridyl azine ligands-a combined experimental and DFT approach
Synthesis, crystal structure characterization and computational investigation of new thieno[2,3-b]pyridine derivatives as potent against molecule p38 alpha MAP kinase
Synthesis, Crystal Structure, and Theoretical Calculations of a μ-Dichloro-Bridged Tetranuclear Cu(II) Bis(salamo)-Type Complex
Synthesis, crystal structure, fluorescence properties, and theoretical studies of a dinuclear Ni(II) complex derived from a quinoline-containing half-salamo-type ligand
Synthesis, Crystal Structure, Fluorescence Property, and Theoretical Investigation of Counteranion-Introduced Ni(II) Complex with Pyridine-Appended Half-Salamo-Like Ligand
Synthesis, Crystal structure, Hirshfeld surface analysis and DFT calculations of 1,2-bis(1-phenylethylidene)hydrazine
Synthesis, crystal structure, Static and dynamic properties, molecular structure, reactive sites, wavefunction and molecular docking of 1-(3-((4-(diethylamino)-2-hydroxybenzylidene)amino)phenyl)ethan-1-one
Synthesis, DFT, ADME and Docking Studies of Homoegonol and Egonol as Potential Inhibitors of COVID-19 Main Protease (6LU7)
Synthesis, functional group analysis (experimental and theoretical), Solvent –Solute interactions, structural insights of (E)-3-(4-chloro-3-(trifluoromethyl) phenyl) imino)
Synthesis, in-Silico investigations, molecular docking, ADMET, and anti-lung cancer activity studies of 1,2,4,5-tetraazaspiro [5.5] undecane-3-thione
Synthesis, molecular structure, Experimental and theoretical characterization of 3-((2-(2,4-dinitrophenyl) hydrazone) methyl) pyridine- Carcinopreventive activity (In Silico and In Vitro Investigation)
Synthesis, NoSpherA2 refinement, and noncovalent bonding of abiraterone bromide monohydrate
Synthesis, SC-XRD structure, spectroscopy, intermolecular interactions, DFT TD-DFT investigation, and (static, dynamic) NLO properties of (2E,5Z)-3-(4-fluorophenyl)
Synthesis, solvation effects, spectroscopic, chemical reactivity, topological analysis and biological evaluation of 4-chloro-N-(2, 6-dichlorobenzylidene) benzohydrazide
Synthesis, structure, theoretical studies, and properties of dinuclear Cu(II) and novel trinuclear Co(II) complexes with a more flexible 3-MeOsalamo-like ligand
Systematic Engineering of Properties of Graphene Quantum Dots by Aryl Amines
Systematic separation of carboxylic acids from H8[PMo7V5O40]-catalyzed lignite oxidation products via deep eutectic solvent
Tailored Design of π-Extended Multi-Resonance Organoboron using Indolo[3,2-b]Indole as a Multi-Nitrogen Bridge
Tailoring intra-molecular coupling in BDT based copolymers to enhance their performance in fullerene-free Organic solar cells
Tailoring the adsorption behaviors of flucytosine on BnNn (n = 12, 16, 20, and 24) nanocage scaffolds
Ternary choline chloride-benzene sulfonic acid-ethylene glycol deep eutectic solvents for oxidative desulfurization at room temperature
Tetracoordinate Co(II) Complexes with Semi-Coordination as Stable Single-Ion Magnets for Deposition on Graphene
The Adsorption of Sulfur Mustard Chemical Warfare Agent on the Ga12N12 and Ca12O12 nanocages; A systematic DFT study
The capture performance of An-MOF for fission gases-Insight from DFT and AIMD calculations
The effect of height and disclination angle of boron nitride nanocones on electronic and structural properties
The effect of interactions on the sensitivity of Csingle bondNO trigger bonds in FOX-7
The Effect of Resonance-Assisted Hydrogen Bond on the Second-Order Nonlinear Optical properties of Pyridine Hydrazone Photoswitches
The Effects of Structural Variations in Neutral Phosphorus Extractants on UO22+ Extraction Behavior and Radiation Stability
The electronic properties, stability and catalytic activity of metallofullerene (M@C60) for robust hydrogen evolution reaction-DFT insights
The experimental and theoretical spectroscopic elucidation of molecular structure, electronic properties, thermal analysis, biological evaluation, and molecular docking studies of isococculidine
The extraction of aromatics using n-methylpyrrolidone-Liquid-liquid equilibrium determination and mechanism exploration
The geometrical isomer effect on the structures and photophysical properties of small molecular fluorophores
The influence of proton-donor solvent on [4+2] benzannulation reaction of dienal with tertiary enaminone
The Linkage-Moderated Covalent Organic Frameworks with -C=N- and -N=N- on Charge Transfer Kinetics Towards the Robust Photocatalytic Hydrogen Activity
The methylene blue and phenol red photocatalytic degradation study by cyclodextrin-ZnO nanostructures
The role of ion-membrane interactions in fast and selective mono-multivalent ion separation with hierarchical nanochannels
The role of trifluoroacetic acid in new particle formation from methanesulfonic acid-methylamine
The Role of Water in the Adsorption of Nitro-Organic Pollutants on Activated Carbon
The roles played by anionic and cationic species in the curing process of aromatic nitrile resins
The scandium organometallic behavior. Design of a chamber of reaction to trap CO2
The Study of DFT and TD-DFT Series of Guaiazulenic Substituted Azobenzene Containing Chromophores for DSSC Materials
The Unconventional Noncovalent Interactions Control-Crystallographic and Theoretical Analyses of the Crystalline Structure of 1,1′-(1-Chloro-4-methoxyphenyl)dibenzene as a Case Study
The Unique Ambiphilicity of Tellurium in the [MesitylTe(I)(I2)(I3)]- Anion
Theoretical and experimental analysis of CO2 capture into 1,5-diamino-2-methyl-pentane n-propanol non-aqueous absorbent regulated by ethylene glycol
Theoretical and experimental cocrystal screening of temozolomide with a series of phenolic acids, promising cocrystal coformers
Theoretical and experimental studies on the electrodeposited nanocrystalline Ag coatings from a novel Ag2O-based non-cyanide electrolyte
Theoretical Approach towards Benzodithiophene-Based Chromophores with Extended Acceptors for Prediction of Efficient Nonlinear Optical Behaviour
Theoretical aspects of the adsorption of normal and modified base pairs of DNA on graphene models toward DNA sequencing
Theoretical calculations and electrochemical investigation of additives in aqueous methanesulfonic acid for lead electrodeposition
Theoretical design of transition metal-doped oxo-triarylmethyl as a disposable platform for adsorption of ibuprofen
Theoretical exploration of bromine substitution effect and hydrostatic pressure responsive mechanism for room temperature phosphorescence
Theoretical implications on the [3?+?2] cycloaddition reactions of dibromoformaldoxime and (Z)-, (E)-3-(4-chlorobenzylidene)-1-methylindolin-2-one in terms of FMO, MEDT, and distortion-interaction theories
Theoretical insight into dimethyl carbonate carboxymethylation of alcohols assisted by Lewis acid proton carrier catalyst FeCl3
Theoretical insight into the activity and selectivity in palladium-Ming-Phos-catalyzed three-component asymmetric synthesis of gem-diarylmethine silanes
Theoretical insight on Cm(III) and Eu(III) competing with Am(III) for binding to N-donor extractants with different dentate numbers
Theoretical insights into photo-induced behavior for 3-(1H-phenanthro[9,10-d]imidazol-2-yl)-9-phenyl-9H-carbazol-4-ol fluorophore
Theoretical insights into the structure, stability, thermochemistry, and bonding in hydrated N2O clusters
Theoretical investigation of anion perfluorocubane
Theoretical Investigation of Single-Atoms Encapsulated by Fullerenes (C59X X=As, Ga, Ge) as Biosensors For Uric Acid (UA)
Theoretical investigation of the CO oxidation mechanism over Mn-corrole as a Single Atom Catalyst
Theoretical Investigation on Solvents Effect in Molecular Structure (TD-DFT, MEP, HOMO-LUMO), Topological Analysis and Molecular Docking Studies of N-(5-((4-Ethylpiperazin-1-yl)Methyl)Pyridin-2-yl)-5-Fluoro
Theoretical investigation on switchable [3 + 3] cycloaddition of vinyl sulfoxonnium ylide with cyclopropenone for divergent synthesis of phenol
Theoretical investigation on the structural properties, scavenging mechanism and toxicological profile of Nymphaeol C, a flavanone in Macaranga Tanarius
Theoretical investigation, solvation effect, topology, NCI and molecular docking studies on potential Pemigatinib
Theoretical Investigations of Dye-Sensitized Solar Cells
Theoretical Investigations of Palladium-Catalyzed [3+2] Annulation via Benzylic and meta C?H Bond Activation
Theoretical investigations on structural, spectral, NBO, NLO and topology exploration (AIM, ELF, LOL, RDG) of piperazine-2,5-dione oxalic acid monohydrate
Theoretical Modeling of B12N12 Nanocage for the Effective Removal of Paracetamol from Drinking Water
Theoretical Perspective toward Designing of 5-Methylbenzo [1,2-b 3,4-b′ 6,5-b″] trithiophene-Based Nonlinear Optical Compounds with Extended Acceptors
Theoretical probe into the universal tetrad effect for the complexation discrepancy between lanthanide elements and organophosphoric extractant diethyl phosphoric acid
Theoretical studies on the interaction of HXeF with M3N3H6 (M = B, Al and Ga) and their substituted derivatives
Theoretical study of new 3-(methylthio)-8-phenyl-8H-thieno[2,3-b]indole derivatives for application in DSSC
Theoretical study of novel oxazine energetic compounds-enthalpy of formation, detonation performance, and sensitivity
Theoretical study of the reaction of borata derivatives of benzene, anthracene and pentacene with CO2
Theoretical study of the saturation and nature of the hydrogen bonds to gold
Theoretical study on fluorescent T-analogues for selective recognition of 2-aminoadenine (Z) in aqueous solution via base-pair induced ESICT mechanism
Theoretical study on intra-molecule interactions in TKX-50
Theoretical Study on Porphyrin Arch-Tapes of Carbonyl-Inserted Seven-Membered Ring with High Nonlinear Optical Properties
Theoretical Study on the Coordination and Separation Capacity of Macrocyclic N-Donor Extractants for Am(III)-Eu(III)
Theoretical study on the excited state mechanism of the fluorescent probe for detecting HOCl
Theoretical study on tin oxide surface chemistry mechanism and thermodynamic properties for atomic layer deposition equipment optimization
Thermal annealing promoted room temperature phosphorescence-motion models and internal mechanism
Thermal stability and thermal decomposition mechanism of octamethyltrisiloxane (MDM)-Combined experiment, ReaxFF-MD and DFT study
Thermally Activated Delayed Fluorescence Emitters Based on a Special Tetrahedral Silane Core
Thermally activated delayed fluorescence tetradentate ligand-containing gold(III) complexes with preferential molecular orientation and their application in organic light-emitting devices
Thermo-activated periodate oxidation process for tetracycline degradation-Kinetics and byproducts transformation pathways
Thermodynamic investigation on ion structure and conductivity of LiF–NdF3 molten salt
Thiophene-Containing Covalent Organic Frameworks for Overall Photocatalytic H2O2 Synthesis in Water and Seawater
Three heteroleptic copper(I) complexes with [Cu(P?P)N2]+ structure and their fluorescence sensing for VOCs
Three-Center M–H–B Bonds Are Strong Field Interactions. Synthesis and Characterization of M(CH2NMe2BH3)3 Complexes of Titanium, Chromium, and Cobalt
Time-resolved excited-state dynamics of star-shaped carbazole-based room temperature phosphorescent molecule by ultrafast absorption spectroscopy
Topological analysis and reactivity study of monomeric and dimeric forms of 2-methyl-4(1H)-quinolone
Topological analysis of information-theoretic quantities in density functional theory
Topology analysis of six phytochemicals through ELF and LOL basins – A DFT study
Torsion Angles between Donor and Acceptor Moieties as a Descriptor for Designing Nonlinear Optics and Thermally Activated Delayed Fluorescence Materials
Touching the density limits of energetic materials by molecular design
Toward a molecular understanding on conductivity of lithium-ion conducting polyanions polymer electrolytes by molecular dynamics simulation
Towards red thermally activated delayed fluorescence emitter-Molecular design and property prediction of phenothiazine-triphenyltriazine derivative
Transformation behavior of the ultraviolet absorbent 2-(2H-benzotriazol-2-yl)-4,6-di-tert-pentylphenol (UV328) in polyethylene plastic under chlorination
Transmittal Effect Evaluation of Heterocyclic Rings on Nonlinear Optical Ambit of Benzotrithiophene-Based Push-Pull Driving Materials-a Theoretical Approach
Trianglimine-Mediated Selective Sieving of Cis Isomer from the Mixture of Dihaloethenes-A Combined Molecular Dynamics and DFT Investigation
Tunable ESIPT pathways in 3-hydroxychromone derivatives
Tuning electron delocalization of hydrogen-bonded organic framework cathode for high-performance zinc-organic batteries
Tuning of the Singlet–Triplet Energy Gap of Donor-Linker-Acceptor Based Thermally Activated Delayed Fluorescent Emitters
Tuning the reactivity of TiO2 layer with uniform distribution of Sub-5 nm Fe2O3 particles via in situ voltage-assisted oxidation for robust catalytic reduction - 快捷方式.lnk
Turn on the luminescence channel by the suppression of conical intersection process for the sulfur-containing hydrogen-bonded molecules
Two different coordination polyhedra in the crystal structure of a novel Zn(II) hybrid complex-DFT investigation, biological activities and emission properties
Two novel quinoline-decorated half-salamo-type Co(II) complexes-Synthesis, crystal structure, Hirshfeld surface analysis, DFT calculation and fluorescence properties
Two-Photon Absorption Response of Functionalized BODIPY Dyes in Near-IR Region by Tuning Conjugation Length and Meso-Substituents
Ultrahigh-printing-speed photoresists for additive manufacturing
Unambiguous discrimination of all 20 proteinogenic amino acids and their modifications by nanopore
Uncovering dissolution behavior and thermodynamic properties of metronidazole benzoate in twelve mono-solvents by experiments and molecular simulation
Uncovering the effects of different substituents on excited state hydrogen-bonding interaction and fluorescent behavior in BTN system
Undercovering the role of physicochemical properties of amides in two-phase absorbent systems for ship exhaust pollutant removals
Understanding adsorption mechanism of a novel “Y” structure carboxylate-hydroxamate surfactant towards rhodochrosite
Understanding of electronic, structural and optical properties of transition metal doped C 20 nano-cage
Understanding the Electronic Structure of the Alkaloid in Scorpion Venom (ASV) Through Drug Adsorption and Molecular Docking Studies on COVID-19 Proteins
Understanding the Influence of Functional Groups on the PEO Composite Electrolytes through a Theoretical, Spectroscopic, and Conduction Behavior Study of Model Systems
Understanding the Reaction Mechanism of Ni-Catalyzed Regio- and Enantioselective Hydroalkylation of Enamines
Unraveling the Aromatic Rule of Cyclic Superatomic Molecules in π-Conjugated Compounds
Unraveling the Role of Silyl and Silane in Si–Ni Catalysts for Hydrogenation
Unraveling the structural and dynamic heterogeneities of hydroxyl-functionalized di-cationic ionic liquids (HFDILs)
Unravelling the structural features of monosaccharide glyceraldehyde upon mono-hydration by quantum chemistry and rotational spectroscopy
Unveiling enhanced electron-mediated peroxymonosulfate activation for degradation of emerging organic pollutants
Unveiling the adsorption and inhibition mechanism of thiadiazole derivatives for mild steel corrosion in hydrochloric acid based on experimental approaches and first-principles calculations
Unveiling the correlation between catalytic efficiency and acidity of metal-free catalyst in hydrogenation reaction
Unveiling the mechanism, regiochemistry and substituent effects of the [3?+?2] cycloaddition reactions
Unveiling the Potential and Mechanisms of 3,3’-Bicarbazole-Based Quaternary Ammonium Salts as Levelers
UV-Light-Induced Morphological Transformation of Spiropyran Assemblies from Irregular Sheet-like Structures to Nanospheres
Verbenone-based push–pull chromophores with giant first hyperpolarizabilities-A new structure–property correlation study
Vibration Spectrum Calculation and Analysis
Vibrational dynamics, Hirshfeld surface and molecular docking studies by quantum computational analysis of 3-Hydroxy-4-nitrobenzaldehyde
Vibrational spectroscopy and density functional theory study of sulfamethoxazole
Vibrational spectroscopy of neutral and cationic 2-Methoxyethanol
Virtual assessment achieved two binary cocrystals based on a liquid and a solid pyridine derivative with modulated thermal stabilities
Visible-light-driven photocatalytic degradation of a typical UV filter by a novel composite photocatalyst
Visible-Light-Driven [2 + 2] Photocycloaddition for Constructing Dimers of N,N′-Diacyl-1,4-dihydropyrazines
Visualization of production and remediation of acetaminophen-induced liver injury by a carboxylesterase-2 enzyme-activatable near-infrared fluorescent probe
Water-Assisted Programmable Assembly of Flexible and Self-Standing Janus Membranes
Weak interaction-steered evolution of polyoxovanadate-based metal-organic polyhedra from transformation via interlock to packing
Why is the ground state of m-benzoquinone triplet
Working mechanism of MXene as the anode protection layer of aqueous zinc-ion batteries
Zn Ionophores to Suppress Hydrogen Evolution and Promote Uniform Zn Deposition in Aqueous Zn Batteries
Zn2+-Induced aggregation of a water-soluble iridium complex enhances aggregation-induced emission for intracellular Zn2+ imaging
Zwitterionic polymers as high-performance coatings for hemoperfusion adsorbents and their chemisorption of protein-bound toxins through computational simulations
[(K,Rb)@([2.2.2]crypt)]2(K,Rb)4[Si9W(CO)4] · 13.4 NH3 – The First Tungsten Functionalized Silicon Zintl Cluster
[Cs?C18]+ and [Na?C14]+ perfect planar alkaline-metal-centered polyynic cyclo[n]carbon complexes with record coordination numbers
[Fe(bpy)Cl3X][bpy H] complexes-synthesis, characterization and theoretical studies towards visible-light photocatalytic hydroxyethylation of quinoline
“Probing the Effect of Acceptor Moiety Engineering in Carbazole-Based Hole-Transporting Materials for Efficient Perovskite Solar Cells”
未恰當引用Multiwfn的文章:
28π Non-Fused Hexaphyrin-Synthesis, Protonation Triggered M?bius Aromaticity and Structural Properties
A Green and Efficient Solvent for Simultaneously Leaching Co and Li from Spent Li-Ion Batteries-Dicarboxylated Polyethylene Glycol
A loofah-based all-day-round solar evaporator with phenolic lignin as the light-absorbing material for a highly efficient photothermal conversion
A molecular electron density theory study of asymmetric Diels–Alder [4?+?2] reaction’s mechanism of furan with three substituted alkynes (5-R substituted-3-(3-(phenylsulfonyl)-propioloyl)-oxazolidin-2-one)
A quantum mechanical investigation of nanocone oxide as a drug carrier for zidovudine-AIDS drug
A Selective and Sensitive Hg2+ Aminonaphthalimide-aza-crown-ether Cellular Chemosensor
A supramolecular self-assembly material based on cucurbituril and cationic TPE as ultra-sensitive probe of energetic pentazolate salts
A surface multiple imprinting layers membrane with well-oriented recognition sites for selective separation of chlorogenic acid from Ficus carica L.
A Theobromine Derivative with Anticancer Properties Targeting VEGFR-2-Semisynthesis, in silico and in?vitro Studies
A Transparent, Tough and Self-Healable Biopolymeric Composites Hydrogel for Open Wound Management
Adsorption of pentabromodiphenylether pollutant by metals (Si, Ge, Sn) encapsulated Zn12O12 nanoclusters-a computational study
Adsorption, excitation analysis, and the mechanism of tetracycline photodegradation by Ca12O12-PEDOT, Mg12O12-PEDOT, and Zn12O12-PEDOT hybrid materials
An intelligent solvent-responsive surface molecularly imprinted membrane with switchable adsorption-desorption performance for selective separation
Anaerobic hydrolysis of recalcitrant tetramethylammonium from semiconductor wastewater-Performance and mechanisms
Anti-breast cancer potential of a new xanthine derivative-in silico, antiproliferative, selectivity, VEGFR-2 inhibition, apoptosis induction and migration inhibition studies
Antilymphoma activities of benzo bisthiazole derivative by molecular docking, impact of solvation, quantum chemical study, and spectroscopic (FT-IR, UV, NMR) investigation
Application of engineered heme enzymes based on myoglobin with high peroxidase activity for efficient degradation of various emerging pollutants
Bimetallic capture sites on porous La-Bi hydroxyl double salts for efficient phosphate adsorption-Multiple active centers and excellent selective properties
Binding interaction of environmental DNA with typical emerging perfluoroalkyl acids and its impact on bioavailability
Biochar encapsulated metal nanoflowers for high efficient degradation of metronidazole via peroxymonosulfate activation
Biodegradation mechanism of chlorpyrifos by Bacillus sp. H27-Degradation enzymes, products, pathways and whole genome sequencing analysis
Biomass-based indole derived fluorescence sensor composited with cellulose paper-Detection of picric acid in food and environment samples
Boosting Hydrogenation of Graphene Quantum Dot-Modified Photocatalysts-Specific Functionalized Modulation at Active Sites
Boosting the sodium storage performance of iron selenides by a synergetic effect of vacancy engineering and spatial confinement
Chromogenesis of a black fluoran dye-Theoretical and spectral studies
Comparative analysis of electronic, magnetic, catalytic properties of clusters (PS4, CrnPS4, AlnPS4, GanPS4, n?=?1?~?3) based on density functional theory
Comprehensive quantum chemical study of the associative complex of para-aminobenzoic acid and 7-diethylamino 4-methyl coumarin by adsorption and aromatic bridges
Computational biology-based study of the molecular mechanism of spermidine amelioration of acute pancreatitis
Computational, Solvatochromic Interactions and Biological Efficacy of the Synthesized 2-Aminothiazole Incorporated Mono-Azo Molecules
Constructing fluorescence probes with fluoride-substituted donors for detecting ?OH in living cells and zebrafish
Construction and Application of Biobased PDES Ionic Gels with a Soft–Hard Segment
Convergent paired electrolysis for the synthesis of pyrazolyl-substituted tetrahydroisoquinolines
Deep insights into novel hexa-coordinated phenoxy-bridged trinuclear Ni(II) asymmetric salamo-like complexes affected by different types of combined solvents
Density functional study on the catalytic properties of the MMoS4 (M=Ni, Co, Fe) cluster
Design, synthesis, anti-proliferative evaluation, docking, and MD simulation studies of new thieno[2,3-d]pyrimidines targeting VEGFR-2
Designing of dimethylfluorene-based hole transport materials for high-performance organic-perovskite solar cell
DFT exploration-Optical and conduction band switching in indaceno donor moiety and its TiO2 adsorbed complexes for an efficient ON-OFF ratio
Discovery and synthesis of atropisomerically chiral acyl-substituted stable vinyl sulfoxonium ylides
Discovery of new VEGFR-2 inhibitors and apoptosis inducer-based thieno[2,3-d]pyrimidine
Donor–Acceptor Molecules with Locked sp3 Carbon Link for Highly Efficient Near Ultraviolet Organic Light-Emitting Diodes
Double-locked narrowband hybrid locally-excited and charge-transfer emitter exhibiting amplified spontaneous emission
Easy separation dual-function Cu2O@LDH@Fe3O4 adsorbent for the removal of Cr(VI) under dark conditions
Effect of Binding Pockets on the Kinetics and Thermodynamics of Diels-Alder Reaction in Cucurbit-Uril Family
Effect of cyano substitution in TADF molecules on luminescence properties
Effect of π-spacers on quinoxaline centered framework toward efficient organic solar cells
Effects of sunflower pectin on thermal stability of purple sweet potato anthocyanins at different pH
Efficient and stable N-heterocyclic ketone-Cu complex catalysts for acetylene hydrochlorination
Efficient room-temperature phosphorescence with tunable lifetime through light modulation from flexible polymer films
Electrolyte Salts for Sodium-Ion Batteries-NaPF6 or NaClO4
Electrolyte Solvation Regulation Engineering Promotes Li-SPAN Battery without Esters
Elucidating the efficacy of plant-derived triterpenoids for treating urinary tract infections
Enantioconvergent construction of stereogenic silicon via Lewis base-catalyzed dynamic kinetic silyletherification of racemic chlorosilanes
Enhanced Degradation of Micropollutants by Visible Light Photocatalysts with Strong Oxygen Activation Ability
Enhancing CH4 separation from surplus N2 in Al-based metal organic frameworks through liner ligand regulation
Enhancing tetraphenylethene cyclization as photoswitch
Enhancing the storage stability of mulberry anthocyanin extract through ternary complex with whey protein isolate and ferulic acid at neutral pH
Equilibrium solubility, solvent effect, equation correlation, and thermodynamic properties of cyclododecanone in twelve organic solvents
Establishment of a reagent-free three-dimensional electro-Fenton system for high H2O2 production and efficient degradation of Roxarsone
Experimental and computational studies of two binuclear Co(II) and Ni(II) bis(salamo)-like complexes
Experimental and theoretical investigation of structure activity relationship on L-Lysine Monohydrate for antioxidant efficacy
Experimental and theoretical investigations on new organic–inorganic silane-fullerol-α-SiC films with enhanced tribo-corrosion resistance
Experimental and Theoretical Studies on Selective Separation of Nd from La-Ce-Pr Using [Omim][DOP]
Exploring the anticancer properties of a new nicotinamide analogue
Exploring the potential of magnetic persimmon tannins-based bio-adsorbent for Cd (II) ions removal from water
Exploring the potential of single-metals (Cu, Ni, Zn) decorated Al12N12 nanostructures as sensors for flutamide anticancer drug
Exploring the Second-Order Polarizability of Copper Doped Silicon Carbide Nanocluster-Toward a New NLO Material
Exploring the Subsistence of Solvation Consequences of l-Asparagine and l-Glutamine Prevailing in Aqueous Ionic Liquid Solutions by Physicochemical and Computational Investigations
Ferrierite nanosheets with preferential Al locations as catalysts for carbonylation of dimethyl ether
Functionalized boron doped graphene (BGP) as smart nanocarrier for delivery of Hydroxyurea (HU) drug
Guest Adaptative Supramolecular Sensing Strategy for Warning the Risky Aflatoxins in Contaminated Cereals
Harmonic Oscillator Model of Aromaticity for Organometallics Containing the Os–C Bond
Heterogeneous catalytic reactions of in-situ generated bromide ions via hydrodehalogenation of tetrabromobisphenol A in advanced oxidation processes over palladium nanoparticles
High-Efficiency and Low-Energy-Loss Organic Solar Cells Enabled by Tuning the End Group Modification of the Terthiophene-Based Acceptor Molecules
Highlight the plasma-generated reactive oxygen species(ROSs)dominant to degradation of emerging contaminants based on experiment and density functional theory
Highly conductive three-dimensional metal organic frameworks from small in situ generated ligand
Identification of CH3F, CH3Cl, and CH3Br molecules by boron phosphide nanosheets-a DFT study
Improving the Cycle Performance of Porous Schiff-Base Polymer Electrode Materials in Lithium-Ion Batteries by Prelithiation
Insight into the effect of microplastics on photocatalytic degradation tetracycline by a dissolvable semiconductor-organic framework
Investigation of manganese-iron oxide nanocomposite immobilized on powdered activated carbon as an efficient activator of peroxymonosulfate for antibiotics degradation
Investigation on the solvent and temperature effect of climbazole solubility
Leveraging the Relative Strengths of Hydrogen and Halogen Bonds to Control Nanostructures
Magnetic surface molecularly imprinted polymers for efficient selective recognition and targeted separation of daidzein
Mechanically Ultra-Robust Elastomers Integrating Self-Healing and Recycling Properties Enable Information Encryption and Hierarchical Decryption
Mechanistic study on levoglucosan condensation and decomposition in the condensed phase during cellulose pyrolysis
Metal doping fabricated heterobimetallic nickel-zinc composites and its performance-enhancing sensitivity towards nitrogen dioxide
Metals (B, Ni) encapsulation of graphene-PEDOT hybrid materials for gas sensing applications-A computational study
Mild and Efficient Method for the In Situ Preparation of High-Quality MXene Materials Enabled by Hexafluoro Complex Anion Contained Salts Etching
Mn(III)–Salen Complexes with Metallophilic Interactions
Mn-doped Carbon-Al2SiO5 Fibers Enable Catalytic Ozonation for Wastewater Treatment
MnCe@HKUST-1 for Efficient Removal of Gaseous Thallium Insights from Kinetic and Experimental Studies
Molecular design of multiple ligand metal–organic framework (ML-MOF) collectors for efficient flotation separation of minerals
Molecular modeling of the structural, electronic, excited state dynamic, and the photovoltaic properties of the oligomers of n-corannulene (n = 1–4)
Multispectral method combined with molecular modelling to investigate the binding mechanisms of DBP and DIBP on pepsin
Naphthalenediimide-Iodobismuthate Hybrid Heterostructures-Water Resistance and Long-Lived Charge-Separated States
Narrowband blue emitter based on fused nitrogen-carbonyl combination with external quantum efficiency approaching 30%
New thiazolidine-2,4-diones as effective anti-proliferative and anti-VEGFR-2 agents
Non-noble-metal TiC nanoparticles promoted charge separation and photocatalytic degradation performance on Bi2O3 microrods
Noncovalent modification induced 2D planar stacking-a promising strategy for high-energy insensitive materials
Nonfluorinated Antisolvents for Ultrastable Potassium-Ion Batteries
Novel low energy mixed amine biphasic solvent for carbon dioxide capture in biogas upgrading
Novel Molecular Insights into the Effects of Ethoxy Group Number on Emulsification and Viscosity Reduction of Anionic–Nonionic Surfactants
Organocatalytic Asymmetric [4 + 2] Cyclization of Azadienes with Azlactones
Oxidative esterification of 5-hydroxymethylfurfural to dimethyl 2,5-furandicarboxylate over Au-supported poly(ionic liquid)s
Partial Methane Oxidation to Methanol on Ru-Porphyrins – on the Role of Non-Innocent Ligands and Spin Crossover
Phosphorus encapsulated gallium nitride and aluminum nitride nanotubes as nonenzymatic sensors for fructose, glucose, and xylose sugars as biomarkers for diabetes-mellitus
Photo and Temperature Responsive Novel Surface Active Ionic Liquid-Based Polymeric Hydrogel
Poly(ether imide) Nanocomposites with BaTiO3@TiO2@SiO2 or BaTiO3@SiO2@TiO2 Fillers Improve Energy Storage Capacity and Dielectric Thermal Stability
Porous cyclopentadiene unit-incorporated graphitic carbon nitride nanosheets for efficient photocatalytic oxidation of recalcitrant organic micropollutants in wastewater
Prediction of Cosolvent Effect on Solvation Free Energies and Analysis of Intermolecular Forces of Tizanidine Hydrochloride in Ethyl acetate + Methanol - Ethanol Mixtures
Preparation of Random Polymers by Copolymerization of Rigid-Twisted Spirobiindane with Poly(aryl isatin) for High-Performance Anion Exchange Membranes
Quantum Computational, Spectroscopic Characterization, Hirshfeld Analysis & Molecular docking Studies on p-Toluenesulfonic Acid (p-TSA) or Tosylic Acid
Ratiometric quantification and visual detection of sulfur dioxide residues using a coumarin-derived fluorescent probe
Reaction Mechanism of Nitrogen-Containing Heterocyclic Compounds Affecting Coal Spontaneous Combustion
Recovery of rare earth metal oxides from NdFeB magnet leachate by hydrophobic deep eutectic solvent extraction, oxalate stripping and calcination
Recyclable sericin-nanocellulose aerogel with high adsorption capacity for tetrabromobisphenol A in water-Insight from DFT calculations
Red mud-based biochar activated peroxydisulfate under visible light for tetracycline removal-Synergistic effect of FeAl2O4 photocatalysis
Removal of As(Ⅲ) and As(Ⅴ) from mine groundwater using bimetallic Fe-Cu nanoparticles
Removal of Microcystis aeruginosa and its extracellular organic matters by using covalently bonded coagulant
Robust Single-Supermolecule Switches Operating in Response to Two Different Noncovalent Interactions
Self-Assembly of Hydrogen-Bonded Organic Crystals on Arbitrary Surfaces for Efficient Amplified Spontaneous Emission
Sensing applications of graphitic carbon nitride (C6N8) for Nitrogen oxides-A DFT study
Sensing of H2S, NO2, SO2, and O3 through pristine and Ni-doped Zn12O12 nanocage
Separation of 1-methoxy-2-propanol and water by organic solvent extraction based on quantum chemistry calculation and liquid-liquid equilibrium experiment
Solvent impact on the photochemical properties of anticancer drug PLX4720; a computational study
Solvent-promoted selective hydroxymethylation of biomass-derived furan compounds
Stability, electronic and catalytic properties of ConMoP(n?=?1?~?5) clusters-A DFT study
Strategies for promoting the degradation of phenol by electro-Fenton-Simultaneously promoting the generation and utilization of H2O2
Structural, electronic features, photoluminescence property, antimicrobial activity and spectral properties of 5-bromosalicylaldehyde derivative
Structure–Property Relationships of ITIC-Based Acceptor–Donor–Acceptor Type Molecules as Novel Organic Nonlinear Optical Materials
Studies on glutathione conjugation reactions of some cyclic chalcone analogs to seek reactivity-biological activity relationships
Study on Dynamic Column Behavior and Complexation Mechanism of DBS-Modified Crown Ether-Based Silica to 90Sr
Study on the Behavior of Saturated Cardanol-Based Surfactants at the Crude Oil-Water Interface through Molecular Dynamics Simulations
Study on the effectiveness of ionic liquid-based biphasic amine solvent in removing H2S, NH3 and CO2 from biogas and its influential characteristics
Study on the performance of MnOx modified graphite felts as electrodes for iron-chromium redox flow battery obtained by permanganic acid etching
Study on the zinc porphyrins as potential carbonic anhydrase mimics for promoting CO2 absorption in K2CO3 solution
Successively separating C3H6 and C2H4 from C2H4-C2H6-C3H6-C3H8 mixture in tandem fixed beds involving two zeolites
Synergistic Additives Enabling Stable Cycling of Ether Electrolyte in 4.4 V Ni-Rich-Li Metal Batteries
Syntheses and Characterizations of Hetero-Bimetallic Chromium-Dinitrogen Transition-Metal Complexes
Synthesis and Photophysical, Electrochemical, and DFT Studies of Piperidyl and Pyrrolidinyl Chalcones
Synthesis of dual p-n heterojunction of NiWO4-CdS-ZIF-67 photocatalyst as a highly efficient photo-assisted fenton-like catalyst for removal of doxycycline hydrochloride
Synthesis of environmentally friendly neonicotinoid insecticide with proper functional properties by theoretical methodologies
Synthesis, characterization and properties of new heat resistant energetic materials based on two C–C bridged pyrazole and benzene skeletons
Synthesis, crystal structure, spectral analysis, DFT calculations, docking studies, in vitro biological activity evaluation and in silico drug-likeness prediction of a novel L-xylose derivative
Synthesis, GCMS, Spectroscopic, electronic properties, chemical reactivity, RDG, topology and biological assessment of 1-(3,6,6-Trimethyl-1,6,7,7a-tetrahydrocyclopenta[c]pyran-1-yl)ethanone
Synthesis, structural characterization and DFT calculations of a diphenyl-2-pyridylphosphine complex of copper(II)
Synthesis, Structural, Third-order Nonlinear Optical Properties and Hirshfeld Surface Analysis of 1-((E)-(4-chlorophenylimino)methyl)naphthalen-2-ol
Synthesis, X-ray Crystallography, spectroscopic Analysis, DFT Calculations, Hirshfeld surface Analysis, and evaluation of Electric, Dielectric, and cytotoxic properties of a new hybrid compound
Tailoring Zn-based diacidic functionalization of Deep eutectic solvent Catalyst-Green and efficient synthesis of ε-Caprolactam under mild conditions
Ternary Mixture of Pentanamide in Solvent Analogy with Halogenated Phenol Experimental, Theoretical, and In Silico Biological Studies
The mechanism of self-assembly of lignin in deep eutectic solvent based on sulfamic acid and urea through molecular dynamics simulation
The regulation of protein crystallization using choline chloride precipitant and its mechanism
Theoretical design and evaluation of efficient small donor molecules for organic solar cells
Theoretical Insights into the Effect of Conjugated Cores on Optical and Photovoltaic Properties of Small Molecule Acceptors for Organic Solar Cells
Theoretical investigation on the influence of electron-accepting substitutions in modulating the optoelectronic properties of dye-sensitized solar cells
Theoretically designing and evaluation of highly efficient small donor molecules for organic solar cells
Thiophene-Rich Benzoxazines with an Amide Moiety-Integration of Structural and Hydrogen Bonding Influence on the Polymerization Mechanism by Experimental and Computational Studies
Transformation of organic afterglow mechanism from room-temperature phosphorescence to thermally-activated delayed fluorescence in intramolecular charge transfer systems
Tuning the reactivity of TiO2 layer with uniform distribution of Sub-5 nm Fe2O3 particles via in situ voltage-assisted oxidation for robust catalytic reduction
Ultrafast underwater self-healing piezo-ionic elastomer via dynamic hydrophobic-hydrolytic domains
Unraveling the Impact of Polar Solvation on the Molecular geometry Spectroscopy (FT-IR, UV, NMR), Reactivity (ELF, NBO, HOMO-LUMO) and Antiviral Inhibitory Potential of Cissampeline by Molecular Docking approach.
Unraveling the single-atom Fe–N4 catalytic site selectivity generate singlet oxygen via activation of persulfate
Utilization of Methyclothiazide Adsorbed with Malonamide for Quantum Chemical applications-A DFT and DFT-D2-D3 Studies
Yttrium- and zirconium-decorated Mg12O12–X (X = Y, Zr) nanoclusters as sensors for diazomethane (CH2N2) gas
Zn-decorated S,P,B co-doped C2N nanosheet for ibuprofen adsorption-Experimental and density functional theory calculation
“Novel 2-D Phosphorene-Based Drug Delivery System for Anti-HIV Zidovudine Drug to Enhance the Therapeutic Effects-A First-Principles Based Study”
文/Sobereva@北京科音 2023-Oct-22
非常流行的波函數分析程序Multiwfn(http://www.shanxitv.org/multiwfn)在Linux下的安裝方法在Multiwfn手冊2.1.2節有充分的說明,但我發現老有人不仔細看,或者由于缺乏Linux最基本常識而無法正確follow,還有人因為那是英文就干脆不看而放棄安裝。鑒于此,這里用中文完整、詳細說一下Multiwfn在Linux環境下的安裝。如果對Multiwfn一無所知,參看《Multiwfn FAQ》(http://www.shanxitv.org/452)和《Multiwfn入門tips》(http://www.shanxitv.org/167)。Multiwfn的可執行文件、源代碼和手冊都可以在官網http://www.shanxitv.org/multiwfn中的Download頁面下載。本文介紹的情況適用于目前Multiwfn官網上最新版本。本文里涉及的命令里的空格必須看清楚,眼睛不好就直接從本文中復制粘貼命令。如果仔細看本文還是安裝不成功,可以去Multiwfn論壇http://bbs.keinsci.com/wfn發帖提問,必須把軟件環境和遇到的問題交代得盡可能具體。
安裝Multiwfn非常簡單,本文文字多純粹是因為講解得詳細。
0 關于Multiwfn的普通版與noGUI版
首先要知道Multiwfn的Linux版有普通版和noGUI版兩種,前者在官網的下載頁面上是Multiwfn_[版本號]_bin_Linux.zip形式的文件名,后者在文件名里多了個noGUI字樣。普通版具有完整的功能,但運行時需要有motif圖形庫提供的libXm.so.4文件(這個文件本身是Multiwfn所用的dislin圖形庫所依賴的),否則無法啟動,安裝這個庫需要root權限。然而在超算或者公用的服務器上,普通用戶又拿不到root權限而沒法安裝motif庫。為了解決這個矛盾,Multiwfn在官網上特意提供了noGUI版,由于它在啟動時不需要motif庫,因此也就不需要弄到root權限來安裝它,故普通用戶都可以很容易地安裝。noGUI版的缺點在于沒法使用任何Multiwfn與圖形有關的功能,如無法顯示出圖形界面、沒法繪制各種曲線圖/平面圖/等值面圖、做拓撲分析后無法直接可視化結果等。由于特意放到高性能Linux服務器上運行Multiwfn通常是為了讓Multiwfn跑一些高耗時的任務,它們往往不直接涉及圖像顯示,所以這倒也不會帶來明顯問題。
下面就開始講Multiwfn在Linux下的安裝流程。
1 安裝motif庫(安裝noGUI版直接跳過此節)
如果你用的是Redhat系操作系統,如Redhat Enterprise Linux,CentOS(包括stream)、Rocky Linux、Fedora等,在確保機子能正常訪問Internet的情況下,在終端(即命令行界面里)輸入yum install motif命令即可安裝motif庫。這需要root權限,要么以root方式登錄,要么在普通用戶下用sudo來執行此命令。
如果你無法訪問Internet,可以自行去https://motif.ics.com/motif/downloads下載適合x86_64架構的最新的motif庫,比如https://motif.ics.com/sites/default/files/motif-2.3.4-1.x86_64_0.rpm,放到Linux服務器上后用rpm -i motif-2.3.4-1.x86_64_0.rpm命令手動安裝之。不過這樣安裝往往會提示缺一些亂七八糟的依賴庫,所以還是建議盡量用yum方式安裝。
如果你不知道當前機子里已經裝了motif沒有,運行rpm -qa|grep motif命令,如果找到了,那就不用再裝了。
上面說的都是Redhat系Linux的情況。如果你是Debian系Linux如Ubuntu的用戶,在連著Internet的情況下可以用sudo apt-get install libxm4 libgl1命令安裝motif和相關的libgl1庫。
2 檢查SysV共享內存段
運行cat /proc/sys/kernel/shmmax命令檢查SysV共享內存段的值,數值的單位為字節。有的系統這個值默認得非常小,比如就32MB,當Multiwfn做一些較耗內存的分析、載入較大波函數文件時會崩潰。發現需要增大的話,在/etc/sysctl.conf里加入比如kernel.shmmax = 2000000000然后重啟系統,就可以令上限提升到大約2GB。修改此文件需要有root權限。
3 解壓Multiwfn
去Multiwfn官網上下載同時帶著bin字樣(binary,即已編譯好的可執行文件)和Linux字樣的壓縮包,放在Linux系統下,用unzip [文件名] 命令將之解壓到當前目錄下。圖形界面里也可以在此文件上點右鍵后選擇解壓。
下文假定解壓后的目錄是/home/sob/Multiwfn_[版本號]_bin_Linux/,在里面應當可以看到Multiwfn程序的各種文件。
4 配置~/.bashrc文件
每個用戶主目錄下都有一個.bashrc文件,記錄了每次進入終端后自動執行的命令。由于這是隱藏文件,所以默認情況下看不到。你可以輸入vi ~/.bashrc(看清楚vi后面的空格)用vi編輯器去編輯它,也可以在Linux圖形界面中要求顯示隱藏文件,然后通過操作系統自帶的有圖形界面的文本編輯器去編輯它。在此文件末尾加上以下內容
ulimit -s unlimited
export OMP_STACKSIZE=200M
export Multiwfnpath=/home/sob/Multiwfn_[版本號]_bin_Linux
export PATH=$PATH:/home/sob/Multiwfn_[版本號]_bin_Linux
然后保存文件。
下面解釋一下以上命令的意義。
ulimit -s unlimited用來去除某些操作系統對堆棧內存使用的限制。某些系統默認的限制設置非常腦殘,不這么設一下的話用Multiwfn處理稍微大一點的波函數都會出現Segmentation fault報錯而終止。
export OMP_STACKSIZE=200M用來將OMP_STACKSIZE環境變量設為200M。Multiwfn并行運算是通過OpenMP技術實現的,即計算會分攤到不同的線程上,往往有很多數組要儲存在每個線程的堆棧內存里。每個線程可以用的堆棧內存量上限就是通過OMP_STACKSIZE來設的,以上例子設成了200MB通常夠用了,如果不夠的話會導致計算崩潰。并行核數乘以OMP_STACKSIZE值必須顯著小于物理內存可用量。
Multiwfn目錄下的settings.ini文件記錄了Multiwfn的配置信息。Multiwfn啟動時首先在當前目錄下尋找settings.ini,如果找不到,則會在Multiwfnpath環境變量設定的目錄下找settings.ini,如果還找不到,則會使用默認設定(和程序壓縮包里settings.ini里的原始設置相同)。由于在Linux下啟動Multiwfn時通常不是在Multiwfn目錄下啟動的,這是為什么上面要定義Multiwfnpath環境變量,用來避免Multiwfn找不到settings.ini。
PATH是Linux系統的一個重要的環境變量,里可以包含一大堆目錄。如果將一個可執行文件所在目錄加入其中,那么在任意目錄下都可以直接輸入可執行文件名而不用帶著目錄名就可以啟動之。如上將Multiwfn所在目錄加入PATH環境變量就是為了這個目的。
5 增加可執行權限
Multiwfn的普通版的可執行文件是Multiwfn目錄下的Multiwfn,noGUI版是Multiwfn目錄下的Multiwfn_noGUI。現在給它們加上可執行權限使之可以被運行。
對于普通版,在終端里運行chmod +x /home/sob/Multiwfn_[版本號]_bin_Linux/Multiwfn。如果是noGUI版,顯然把其中的Multiwfn替換成Multiwfn_noGUI。
6 配置settings.ini
編輯Multiwfn目錄下的settings.ini,搜索nthreads,將之數值改為計算時要用的并行核數,通常設為CPU的物理核心數即可。
settings.ini里還有幾個其它設置想改的話可以根據需要修改一下
?formchkpath:定義的是Gaussian目錄下的formchk程序的路徑。formchk是干嘛的在《詳談Multiwfn支持的輸入文件類型、產生方法以及相互轉換》(http://www.shanxitv.org/379)里明確說了。如果把它設成了formchk的實際的路徑,Multiwfn就可以直接載入Gaussian的chk文件(在載入時會自動調用formchk轉換成fch文件后載入)。
?orca_2mklpath:如果設成了ORCA目錄下的orca_2mkl可執行文件的路徑,Multiwfn就可以直接載入ORCA計算產生的gbw文件(在載入時會自動調用orca_2mkl轉換成molden文件后載入)。
?gaupath和orcapath:當它們分別設成了Gaussian和ORCA的可執行文件的路徑,Multiwfn的一些功能才能自動調用Gaussian和ORCA進行計算,如《使用Multiwfn超級方便地計算出概念密度泛函理論中定義的各種量》(http://www.shanxitv.org/484)介紹的功能。
7 測試
退出終端(關閉終端窗口,或者斷開鏈接),然后重新進入終端,之后直接輸入Multiwfn(普通版)或Multiwfn_noGUI(noGUI版)應當就能進入Multiwfn了。之后可以隨便做簡單的測試,比如載入Multiwfn程序包自帶的examples目錄下的文件然后做簡單的計算。例如載入examples/CH3CONH2.fch,之后依次輸入
9
8
屏幕上馬上就會輸出《Multiwfn支持的分析化學鍵的方法一覽》(http://www.shanxitv.org/471)里介紹的拉普拉斯鍵級。
8 其它
如果你是通過純文本界面連接遠程Linux服務器并在上面執行Multiwfn的普通版,并且發現啟動Multiwfn時會短暫卡住,在~/.bashrc文件末尾加入export DISPLAY=":0"可以避免。
《在Linux系統下安裝Multiwfn 3.6的演示(CentOS 7.6)》(https://www.bilibili.com/video/av41402462/)是一個較老版本Multiwfn在Linux下的安裝演示,如果你在follow上文時遇到困難可以參考。里面有些流程和細節與本文不同,以本文為準。
]]>文/Sobereva@北京科音 2023-Aug-11
1 前言
(超)極化率密度是一種非常重要的直觀分析化學體系(超)極化率內在本質的方法,由此可進一步得到三維空間各個位置對(超)極化率的貢獻,這種分解分析對于討論和對比(超)極化率的大小和符號,以及深入了解體系不同區域對外電場的響應,都有重要意義。筆者之前在《使用Multiwfn計算(超)極化率密度》(http://www.shanxitv.org/305)介紹了如何利用強大靈活的波函數分析程序Multiwfn以純手動的方式計算(超)極化率密度,此方法已經在大量文章中得到了廣泛的應用。但這種做法操作起來很費事,計算流程也容易忘,經驗不足的用戶還可能犯錯誤,在一定程度上制約了(超)極化率密度的普及。
2023-Aug-11更新的Multiwfn版本中加入了非常方便省事的計算(超)極化率密度以及三維空間對(超)極化率的貢獻的功能,可以直接繪制等值面圖和平面圖,令這種分析的流程極大地簡化,也充分避免了初學者在操作過程中犯錯的可能,在本文就介紹一下這個功能的用法。《使用Multiwfn計算(超)極化率密度》(http://www.shanxitv.org/305)里介紹的操作方法就可以徹底棄了,但如果你對(超)極化率密度的背景知識不了解、不知道怎么分析的話,則一定要先讀一下這篇文章里的相關介紹和討論,我假定讀者已經看過了此文。
Multiwfn可以在官網http://www.shanxitv.org/multiwfn免費下載。如果你對Multiwfn不了解,建議閱讀《Multiwfn入門tips》(http://www.shanxitv.org/167)和《Multiwfn FAQ》(http://www.shanxitv.org/452)。本文介紹的功能需要結合Gaussian使用,本文例子用的是Gaussian 16 C.02版,至少不能用比G09 B.01更老的版本。
下文涉及的文件都可以在http://www.shanxitv.org/attach/683/file.zip中找到。
2 實例1:D-pi-A體系第一超極化率密度的繪制
此例要對下面這個D-pi-A型體系繪制第一超極化率密度的XX分量(ρxx),以及三維空間對第一超極化率的貢獻(-x*ρxx)。在Multiwfn可執行文件包里examples\excit\D-pi-A.fchk文件記錄的結構信息對應的是此體系的優化過的基態的結構,如下圖所示。
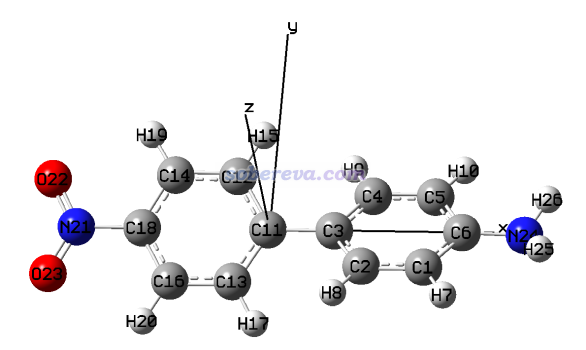
啟動Multiwfn,然后輸入
examples\excit\D-pi-A.fchk //Multiwfn將從中讀取結構信息。用.xyz、.pdb、.gjf等其它記錄了結構信息的文件格式也都可以,詳見《詳談Multiwfn支持的輸入文件類型、產生方法以及相互轉換》(http://www.shanxitv.org/379)
24 //各種(超)極化率相關的分析
3 //(超)極化率密度分析
2 //計算第一超極化率密度
1 //X方向
1 //產生不同電場下的Gaussian單點任務的輸入文件
現在當前目錄下就有了X-1.gjf、X_0.gjf和X+1.gjf。Multiwfn用的電場的有限差分步長是0.003 a.u.,這很適合用于計算(超)極化率密度,也因此比如X-1.gjf和X+1.gjf就分別對應向X的負方向和正方向加0.003 a.u.電場,X_0.gjf是不加外電場的情況。輸入文件里默認用的是PBE0/aug-cc-pVTZ進行計算,這對于大多數分子的(超)極化率的計算都是適合的級別,如果想改成其它的自己改就完了。輸入文件里還有一些關鍵詞是用來盡可能避免因為帶彌散函數較多時電子積分求解精度差和KS矩陣構建不精確而導致SCF難收斂的,不懂就別亂改。nosymm關鍵詞也自動加上了,由此避免Gaussian自動把體系搞到標準朝向下,不理解的話看《談談Gaussian中的對稱性與nosymm關鍵詞的使用》(http://www.shanxitv.org/297)。從gjf文件內容還可以看到這些任務會產生與輸入文件同名的.wfx格式的波函數文件。
使用Gaussian運行這三個.gjf文件,分別得到X-1.wfx、X_0.wfx和X+1.wfx。如果你不知道怎么批量用Gaussian運行一堆gjf文件的話,看《使用Gaussian時的幾個實用腳本和命令》(http://www.shanxitv.org/258)。
接著,在Multiwfn的窗口里輸入2,并輸入這些wfx文件所在目錄(如果是在當前目錄下則直接按回車),它們就都被讀取了。之后看到新菜單,選項的含義都提示得非常明白,可以對(超)極化率密度和三維空間對(超)極化率的貢獻計算格點數據(之后可以繪制等值面圖或者導出cub文件),也可以對它們繪制成平面圖。由于一開始我們選的是第一超極化率和X方向,所以此菜單的選項里的first hyperpolarizability density當前顯然指的是ρxx,而spatial contribution to first hyperpolarizability顯然對應-x*ρxx。
下面繪制ρxx的等值面圖。在Multiwfn里輸入
1 //計算(超)極化率密度的格點數據
3 //高質量格點。不懂格點怎么選的話看《Multiwfn FAQ》(http://www.shanxitv.org/452)的Q39
之后屏幕上給出了積分值(-1.721222 a.u.)。選1后蹦出了ρxx的等值面圖。等值面數值設為2后看到的圖像如下,綠色和藍色分別為正值和負值等值面
關閉窗口后還可以選2導出cub文件,然后可以用《在VMD里將cube文件瞬間繪制成效果極佳的等值面圖的方法》(http://www.shanxitv.org/483)介紹的方法在VMD里繪制出效果更好的等值面圖。
接下來繪制-x*ρxx的等值面圖,輸入
0 //返回
2 //計算三維空間對(超)極化率密度貢獻的格點數據
3 //高質量格點
屏幕上顯示格點數據的積分值為7803.891517 a.u.,這對應基于立方格點積分-x*ρxx算的βxxx值。選1后蹦出了-x*ρxx的等值面圖。等值面數值設為5時看到的圖如下(這里還改成了solid+mesh風格顯示以讓圖更有立體感)。可見圖里大部分地方數值為正,是此體系βxxx很大的主要原因。
選0返回后還可以選3和4分別繪制ρxx和-x*ρxx的平面圖,操作和設置方式與Multiwfn的主功能4繪制平面圖完全一樣,如果你不熟悉用Multiwfn繪制平面圖的話參看Multiwfn手冊4.4節的海量例子。平面圖的繪制在下一節的例子里有具體體現。
在本文的文件包里的D-pi-A目錄里有個gamma.out,是PBE0/aug-cc-pVTZ級別對當前體系計算(超)極化率的輸出文件。使用《使用Multiwfn分析Gaussian的極化率、超極化率的輸出》(http://www.shanxitv.org/231)介紹的方法查看βxxx會發現數值為7639.07。上面基于立方格點積分得到的值7803.89與之很接近,說明以上圖像是合理、有意義的。
3 實例2:18碳環第二超極化率密度的繪制
18碳環(cyclo[18]carbon)是個有意思的體系,筆者對它及其類似物做了大量的研究,匯總見http://www.shanxitv.org/carbon_ring.html。在Carbon, 165, 461 (2020)筆者給出了18碳環環平面上的ρxxx和-x*ρxxx的填色等值線圖,這里來重現一下。優化過的18碳環的結構是本文文件包里的C18目錄下的C18.xyz,如下所示,體系在Z=0的XY平面上
啟動Multiwfn,然后輸入
C18.xyz
24 //(超)極化率分析
3 //考察(超)極化率密度
3 //第二超極化率密度及三維空間對第二超極化率的貢獻
1 //X方向
1 //產生不同電場下的Gaussian單點任務的輸入文件
[回車] //凈電荷為0、自旋多重度為1
當前目錄下產生了X-2.gjf、X-1.gjf、X+1.gjf和X+2.gjf。如我在Carbon, 165, 468 (2020)中證明的,PBE0無法正確描述18碳環的長短鍵交替的結構,因此手動把這4個gjf文件里的PBE0替換成非常適合研究碳環體系的wB97XD。然后用Gaussian計算它們,得到X-2.wfx、X-1.wfx、X+1.wfx和X+2.wfx。
在Multiwfn的窗口里輸入2,并輸入這些wfx文件所在目錄以載入它們。之后繪制ρxxx的平面圖,輸入
3 //繪制(超)極化率密度的平面圖
1 //填色圖
[回車] //用默認格點數
1 //XY平面
0 //Z=0
圖像馬上就蹦出來了。之后關閉圖像,輸入以下命令調節作圖效果
19 //修改色彩變化
8 //藍-白-紅
1 //設置色彩刻度下限和上限
-100,100
4 //顯示原子標簽
12 //深綠色
8 //顯示化學鍵
14 //棕色
-8 //把橫縱軸單位改成埃
-2 //設置X、Y、Z(色彩刻度)軸的標簽間隔
2,2,20
-3 //其它設置
2 //設置X、Y、Z軸標簽的小數點位數
1
1
1
0 //返回
2 //顯示等值線
3 //修改等值線設置
4 //刪除某些等值線(這里把數值很小的刪除,免得圖像太亂)
32-41
4 //刪除某些等值線
1-10
1 //保存并返回
-1 //重新作圖
現在得到下圖,可見效果很好。-x*ρxxx的平面圖也可以以類似方式繪制,選-5返回后,再選擇4 Plot plane map of spatial contribution to second hyperpolarizability然后重復上面的操作即可。顯然,也可以類似上一節的例子選擇相應選項繪制ρxxx和-x*ρxxx的等值面圖。
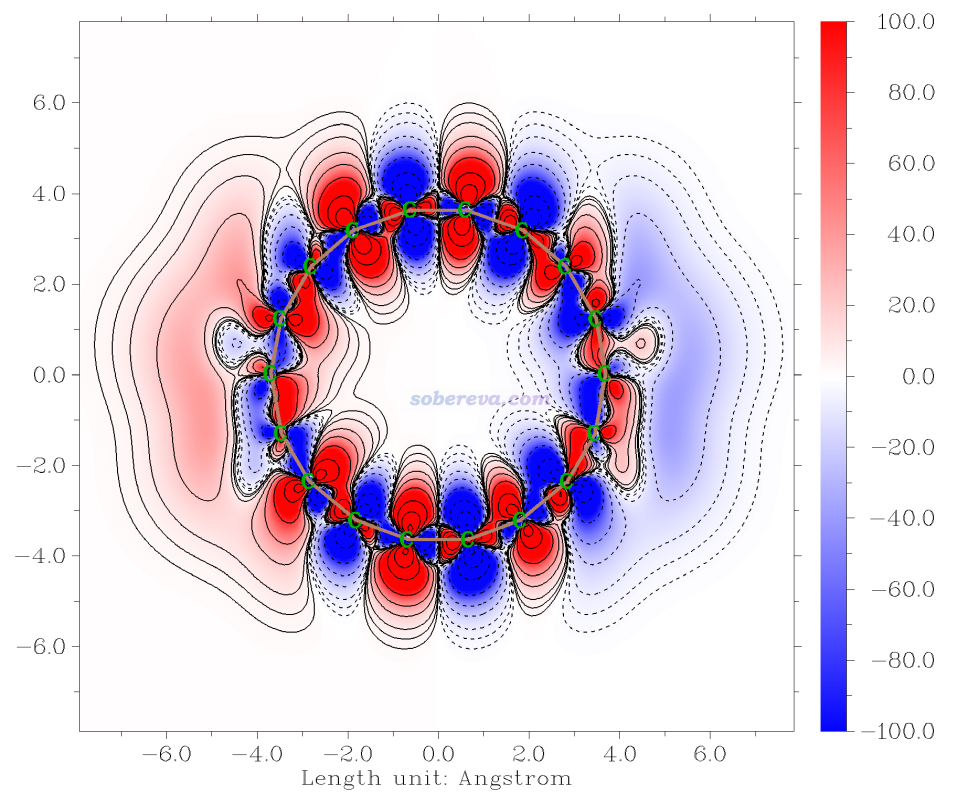
4 總結
本文演示了Multiwfn專門計算(超)極化率密度以及三維空間對(超)極化率貢獻的功能,通過上面的例子可見用此功能繪制等值面圖和等值線圖非常方便,照著屏幕上的提示來操作就可以輕易完成,此功能使得(超)極化率密度的考察變得比以往容易得多得多,推薦大家在極化率和超極化率的研究中充分使用此功能。本文沒有專門給出極化率密度和三維空間對極化率貢獻的計算例子,因為操作和上面的例子完全一樣,只不過進入主功能24的子功能3之后先選擇1 Polarizability density and spatial contribution to polarizability即可。
Multiwfn還可以基于三維空間對(超)極化率貢獻的格點數據通過模糊空間積分來得到原子對(超)極化率貢獻的定量數值,做法在《使用Multiwfn計算(超)極化率密度》(http://www.shanxitv.org/305)的第7節演示了,這里不再重復。怎么把格點數據導出在上面的例子里已經明確提過了。
使用Multiwfn按本文的做法進行研究時需要在寫文章時引用Multiwfn啟動后明確顯示的Multiwfn原文。
]]>文/Sobereva@北京科音 2023-Aug-9
1 前言
NICS在衡量芳香性上用得很普遍,在《衡量芳香性的方法以及在Multiwfn中的計算》(http://www.shanxitv.org/176)里有充分介紹,不了解者務必先閱讀。強大的波函數分析程序Multiwfn具有很好用的繪制一維NICS圖的功能,見《使用Multiwfn繪制一維NICS曲線并通過積分衡量芳香性》(http://www.shanxitv.org/681);此外,Multiwfn支持的ICSS分析在本質上是考察三維空間的NICS(ICSS和NICS僅相差正負號),見《通過Multiwfn繪制等化學屏蔽表面(ICSS)研究芳香性》(http://www.shanxitv.org/216)。雖然如此文所示例的,利用Multiwfn的ICSS模塊結合Gaussian程序產生三維ICSS格點數據后,可以利用Multiwfn以格點數據插值方式得到任意平面上的二維ICSS/NICS圖,但如果你的目的僅僅是繪制二維NICS圖,那么為此很耗時地計算三維ICSS格點數據明顯是不劃算的。本文就演示Multiwfn里新加入的專門繪制二維NICS圖的功能,僅需要借助Gaussian計算分布在作圖平面上的格點即可,耗時比計算三維ICSS格點數據低一、兩個數量級。
讀者請務必使用2023-Aug-7及以后更新的Multiwfn版本,可以在官網http://www.shanxitv.org/multiwfn免費下載。對Multiwfn不了解者建議閱讀《Multiwfn入門tips》(http://www.shanxitv.org/167)和《Multiwfn FAQ》(http://www.shanxitv.org/452)。本文用的Gaussian是G16 C.02。
2 例1:繪制暈苯平面上方1埃處NICS_ZZ平面圖
Multiwfn程序包里的examples\NICS_scan\coronene.pdb是B3LYP/6-31G*級別優化的暈苯的結構文件,如下所示,分子在Z=0的XY平面上。此例繪制分子上方1埃處的平面的NICS_ZZ的填色圖。注意本文里的NICS_ZZ值是指垂直于被考察的環平面的磁屏蔽張量分量的負值,而非特指笛卡爾Z軸方向的分量,后同。
啟動Multiwfn,然后輸入
examples\NICS_scan\coronene.pdb
25 //離域性與芳香性分析
14 //繪制NICS二維平面圖
1 //填色圖
[回車] //用默認的格點數,即兩個方向都是100個點,因此要計算100*100=10000個Bq
0 //設置延展距離
1 //1 Bohr。對當前體系用比默認小得多的延展距離便足以,免得圖中有過多的體系外圍的不感興趣的區域。不知道什么叫延展距離的話看《Multiwfn FAQ》(http://www.shanxitv.org/452)的4.2節
1 //XY平面
1a //Z=1埃
1 //產生Gaussian的NICS二維掃描的輸入文件
examples\NICS_scan\template_NMR.gjf //這是Gaussian做NMR計算的模板文件,里面原子坐標部分用[geometry]代替,會被自動替換
現在當前目錄下產生了NICS_2D.gjf。可以根據實際情況進行修改,比如修改凈電荷、自旋多重度、基組等等,不懂的地方別亂改。這里把基組設為6-31G*,其它的不變。之后用Gaussian計算之,在2*7R32機子96核并行情況下花了1m50s算完。相應的輸入和輸出文件是examples\NICS_scan\目錄下的coronene_NICS_2D.gjf和coronene_NICS_2D.out。
然后在Multiwfn界面里輸入2選擇載入Gaussian輸出文件,然后輸入其路徑examples\NICS_scan\coronene_NICS_2D.out。程序問你要獲得哪種NICS,可以選各項同性值、各向異性值、平行于笛卡爾X或Y或Z方向的分量值、順著特定矢量的分量值。這里選擇5,即平行于Z方向的值。由于當前體系平行于XY平面,因此這么選得到的對應于一般意義的NICS_ZZ。
然后圖像馬上就蹦出來了,同時文本窗口顯示了平面數據最負和最正數值,如下所示,可見作圖平面上NICS_ZZ最負值為-43.3 ppm。可以根據此值恰當地設置色彩刻度上下限范圍
The minimum of data: -43.3034000000000
The maximum of data: 11.3487000000000
關閉圖像,輸入以下命令修改作圖效果
4 //顯示原子標簽
1 //紅色
8 //顯示化學鍵
14 //棕色
17 //設置顯示標簽的距離閾值
5 //距離作圖平面5 Bohr以內的原子標簽才顯示出來
y //更遠的原子用細體字顯示標簽
1 //修改色彩刻度范圍
-45,45
-8 //坐標軸改為以埃為單位
-2 //修改坐標軸刻度
2,2,10
2 //顯示出等值線
3 //修改等值線設置
8 //按等差數列生成等值線數值
-50,5,21 //起始值,步長,步數
y //替換原有的等值線數值。之后等值線數值為-50,-45,-40...略...40,45,50
1 //保存并返回
-1 //重新作圖
現在看到下圖
此圖顏色越深說明NICS_ZZ越負,即對垂直于體系平面方向的磁場屏蔽效應越強。此圖中外圍六元環的顏色明顯比中間的六元環更深,體現出外圍六元環有更強的芳香性。用其它芳香性指標也能體現這一點,比如用Multiwfn計算多中心鍵級的話,會發現中間的環的六中心鍵級是0.022,而外圍的是0.034,也展現出外圍的六元環的芳香性相對更強。
在后處理菜單還可以選-7用來給平面數據乘上一個因子,如果設-1,則數據的符號就會反轉,之后繪制的相當于ICSS平面圖。
3 例2:繪制三苯胺的苯環上方1埃處的NICS_ZZ平面圖
在B3LYP/6-31G*下優化的三苯胺的結構文件是examples\NICS_scan\N(phenyl)3.pdb,如下所示,可見環都是傾斜著的。此例繪制23,24,26,30,28,25原子組成的環(已高亮顯示)的上方1埃平面處的NICS_ZZ平面圖,取垂直于環方向的磁屏蔽張量分量。
啟動Multiwfn,然后輸入
examples\NICS_scan\N(phenyl)3.pdb
25 //離域性與芳香性分析
14 //繪制NICS二維平面圖
1 //填色圖
[回車] //用默認的格點數100*100
8 //繪制某些原子構成的擬合平面的上方或下方的平面
23,24,26,30,28,25 //環中的原子
注意此時在屏幕上顯示了這6個原子擬合的平面的單位法矢量:
The unit normal vector is 0.33076524 0.57300118 0.74984265
接著在Multiwfn里輸入
1 //繪制的是與擬合平面相平行而在它上方1埃的平面。如果輸入負值代表在下方
6 //繪圖平面的邊長為6埃
此時在Multiwfn窗口中顯示了在VMD程序里繪制作圖平面區域的命令:
draw triangle { 2.495 2.581 -1.739} { -1.211 -0.235 2.047} { 6.776 -1.451 -0.547}
draw triangle { -1.211 -0.235 2.047} { 6.776 -1.451 -0.547} { 3.070 -4.266 3.239}
draw material Transparent
如果將N(phenyl)3.pdb載入VMD,然后在文本窗口里輸入以上命令,恰當修改顯示方式后就可以看到下圖,藍色透明區域對應實際作圖平面,由此可以檢驗作圖平面設定得是否合理,當前顯然是合理的。被選擇的六個原子的幾何中心在作圖平面上的投影位置對應于作圖平面的正中心。
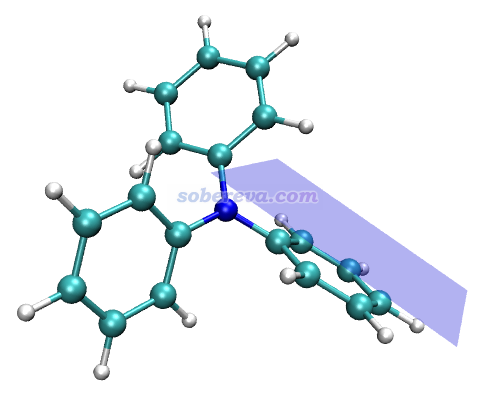
接著在Multiwfn里輸入
1 //產生Gaussian的NICS二維掃描的輸入文件
examples\NICS_scan\template_NMR.gjf //Gaussian做NMR計算的模板文件
當前目錄下產生了NICS_2D.gjf。將基組改為6-31G*,然后讓Gaussian運行,在2*7R32 96核機子上2m5s算完。相應的輸入和輸出文件分別是examples\NICS_scan\目錄下的N(phenyl)3_NICS_2D.gjf和N(phenyl)3_NICS_2D.out。
接著在Multiwfn里輸入
2 //載入Gaussian的NICS掃描的輸出文件
examples\NICS_scan\N(phenyl)3_NICS_2D.out
0 //對各個點,取NICS在特定矢量上的投影
0.33076524 0.57300118 0.74984265 //之前Multiwfn顯示的擬合平面的單位法矢量
之后可見當前的作圖平面上的數據的最負和最正值:
The minimum of data: -34.0268462149550
The maximum of data: 4.05938599881831
關閉圖像,然后輸入
4 //顯示原子標簽
1 //紅色
8 //顯示化學鍵
14 //棕色
17 //設置顯示標簽的距離閾值
5 //距離作圖平面5 Bohr以內的原子標簽才顯示出來
y //更遠的原子用細體字顯示標簽
1 //修改色彩刻度范圍
-34,0
-8 //坐標軸改為以埃為單位
-2 //修改坐標軸刻度
1,1,5
19 //修改色彩變化方式
19 //黃-橙-黑
-1 //重新作圖
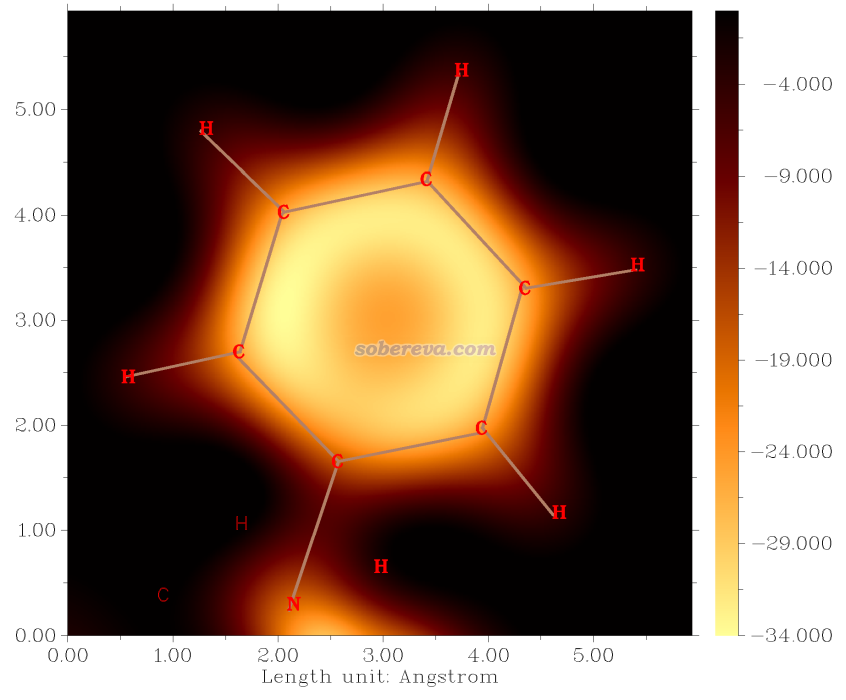
由圖可見在環平面上方的六元環內區域的NICS_ZZ很負,體現出這個環的顯著芳香性。更具體來說,環中心正上方不是最負的,靠近C-C鍵的內側區域是稍微更負的。這樣的NICS分布是普遍現象。
4 例3:繪制C16的垂直于環平面的NICS_ZZ平面圖
筆者之前對18碳環體系(http://www.shanxitv.org/carbon_ring.html)做過大量研究,其中包括其芳香性。其類似物16碳環(C16)最近也被合成了出來。16碳環的in-plane和out-of-plane兩類pi軌道都有16個電子,滿足Huckel反芳香性規則,因此可以預期C16具有雙反芳香性特征。這里使用Multiwfn對垂直于它環平面的截面繪制NICS_ZZ圖,以考察分子由內到外不同區域對垂直于環方向磁場的屏蔽效果。wB97XD/def2-TZVP優化過的C16的結構文件是examples\NICS_scan\C16.pdb,如下所示,可見繪制X=0的YZ平面或者Y=0的XZ平面都可以滿足當前目的。
啟動Multiwfn,然后輸入
examples\NICS_scan\C16.pdb
25 //離域性與芳香性分析
14 //繪制NICS二維平面圖
1 //填色圖
[回車] //用默認的格點數,即兩個方向都是100個點
0 //設置延展距離
1 //9 Bohr。把延展距離設得比默認更大以展現更廣闊區域的磁屏蔽情況
2 //XY平面
0 //Y=0
1 //產生Gaussian的NICS二維掃描的輸入文件
examples\NICS_scan\template_NMR.gjf //Gaussian做NMR計算的模板文件
之后把計算級別改成wB97XD/def2-TZVP,因為筆者在Carbon, 165, 468 (2020)里證明了這個級別可以描述好碳環的結構,而常用的B3LYP則絕對不能用,《談談量子化學研究中什么時候用B3LYP泛函優化幾何結構是適當的》(http://www.shanxitv.org/557)中也提到了這點。而且《我對一篇存在大量錯誤的J.Mol.Model.期刊上的18碳環研究文章的comment》(http://www.shanxitv.org/584)里也說了6-31G*無法合理描述碳環類體系的電子結構,因此這里用的基組質量比前面的例子更好。
此例的NICS掃描的輸入輸出文件是examples\NICS_scan\目錄下的C16_NICS_2D.gjf和C16_NICS_2D.out。在Multiwfn當前的界面里選2并載入此輸出文件,關閉圖像后用以下命令調節作圖設置
4 //顯示原子標簽
12 //深綠色
8 //顯示化學鍵
14 //棕色
17 //設置顯示標簽的距離閾值
50 //距離作圖平面50 Bohr以內的原子標簽才顯示出來
y //更遠的原子用細體字顯示標簽
1 //修改色彩刻度范圍
-50,50
-8 //坐標軸改為以埃為單位
-2 //修改坐標軸刻度
2,2,10
2 //顯示出等值線
3 //修改等值線設置
8 //按等差數列生成等值線數值
-80,5,33 //起始值,步長,步數
y //替換原有的等值線數值
1 //保存并返回
19 //設置色彩變化
8 //藍-白-紅
-1 //重新作圖
看到的圖像如下。可見在環中央及其上下方區域NICS_ZZ都為非常顯著的正值,Multiwfn顯示的平面上最正值都達到了72.3 ppm,說明C16有極強的反芳香性。
上圖可以跟《通過Multiwfn繪制等化學屏蔽表面(ICSS)研究芳香性》(http://www.shanxitv.org/216)里列舉的Carbon, 165, 468 (2020)文中的18碳環截面的ICSS二維圖進行對比,可見特征一樣但符號相反(再次注意ICSS和NICS相差正負號),也即C16和C18的芳香性特征正好是相反的。
5 總結
本文介紹了Multiwfn的非常簡單易用的繪制二維NICS平面圖的功能,定義作圖平面的方式非常靈活,作圖設置豐富,效果極好,在像樣的機子上用Gaussian計算繪制這種圖所需的磁屏蔽張量數據的耗時一般也比較低(在較好的服務器上算不很大的體系屬于立等可取的耗時),非常推薦在研究芳香性問題時恰當地使用。
使用本文的功能繪制NICS平面圖用于論文的情況請按照Multiwfn啟動時的提示恰當引用Multiwfn的原文。
]]>文/Sobereva@北京科音 2023-Aug-6
1 關于NICS曲線及其積分
NICS是一種非常流行的基于磁性質衡量芳香性的方法,在《衡量芳香性的方法以及在Multiwfn中的計算》(http://www.shanxitv.org/176)里有專門的詳細介紹,不了解的話務必先閱讀此文的相關部分。NICS大多數情況是考察特定點(如環中心或環中心上/下方1埃處)的磁屏蔽情況。在J. Phys. Chem. A, 123, 3922 (2019)中作者建議從環中心開始在垂直于環方向上對NICS_ZZ(此處Z指垂直于環方向)進行掃描,并將這一維NICS_ZZ曲線進行積分,文中認為此積分值比諸如NICS(0)ZZ、NICS(1)ZZ那樣只考察個別的點在衡量芳香性上更嚴格和可靠。而且通過考察NICS曲線無疑能比只計算個別位置的NICS獲得更多信息,有助于不同體系間橫向討論和對比。在J. Phys. Chem. A, 126, 3433 (2022)中另外的研究者將這種NICS積分的分析思想應用在氨基酸側鏈苯環的芳香性研究上,并將之稱為integral NICS (INICS)指數。類似于常見的NICS(1)ZZ,垂直穿越某環的NICS_ZZ曲線積分值為明顯負值、接近0、明顯正值說明相應的環表現芳香性、非芳香性和反芳香性。
本文將通過例子介紹如何用強大且易用的Multiwfn波函數分析程序結合Gaussian量子化學程序方便地計算、繪制特定直線上的一維NICS曲線并得到其積分值,也即INICS。無論被考察的環是平行于笛卡爾平面還是歪斜、扭曲的,無論是簡單環狀體系還是結構復雜的體系,此功能都能方便地使用。可以還可以把NICS分解為特定軌道貢獻以了解更深入信息,用于比如分離研究sigma和pi電子對芳香性的貢獻,以及分離考察類似18碳環及衍生物體系(見http://www.shanxitv.org/carbon_ring.html)那樣in-plane和out-of-plane兩套pi軌道對芳香性的貢獻。
Multiwfn之前已支持ICSS分析方法并已得到極其廣泛的應用。ICSS相當于在三維立體空間中計算各個位置的NICS(注意相差正負號),見《通過Multiwfn繪制等化學屏蔽表面(ICSS)研究芳香性》(http://www.shanxitv.org/216)里的詳細介紹。此文也示例了在獲得ICSS三維格點數據后就可以輕易用Multiwfn通過插值方式計算和繪制任意方向上的NICS曲線,但它相對于本文介紹的功能有以下不足:
(1)ICSS格點數據計算相當耗時,沒有像樣服務器的話對于中等大小的體系都算不動,而本文介紹的Multiwfn的一維NICS曲線計算功能在筆記本上也能輕易對不太大的體系完成計算
(2)本文介紹的功能支持NICS分解成軌道貢獻,這是Multiwfn的ICSS模塊不支持的(其實原理上ICSS也能這么分解,只是由于技術原因無法實現:當ghost原子數較多的時候無論用Gaussian自身的AICD接口還是借助NBO 7.0的基于正則分子軌道的NCS分解都會卡住算不動)
(3)ICSS模塊不支持考察任意方向NICS分量,比如垂直于某個傾斜的環方向的分量
另外,Multiwfn還有專門繪制二維NICS圖的功能,見《使用Multiwfn巨方便地繪制二維NICS平面圖考察芳香性》(http://www.shanxitv.org/682)。
下面將結合具體例子講解一維NICS曲線的繪制和積分。讀者務必使用2023-Aug-5及以后更新的Multiwfn版本(注意看程序啟動后顯示的更新日期),Multiwfn可以在主頁http://www.shanxitv.org/multiwfn免費下載。不了解Multiwfn者建議參看《Multiwfn入門tips》(http://www.shanxitv.org/167)和《Multiwfn FAQ》(http://www.shanxitv.org/452)。本文用的Gaussian是G16。
2 例1:計算苯、C5H5-和C7H7+垂直于環的NICS曲線
Multiwfn文件包自帶的examples目錄下的NICS_scan子目錄下的benzene.pdb是在B3LYP/6-31G*級別下優化過的苯的結構,體系處在Z=0的XY平面上。此例對它繪制從環中心下方10埃到環中心上方10埃的NICS曲線,掃描路徑垂直穿越環中心,并且考察的是垂直于環方向的NICS分量(即Multiwfn會讀取Gaussian算的各個掃描點的磁屏蔽張量,取投影出的垂直于環方向的分量。本文后面說的NICS一律都是指取這個分量)。
啟動Multiwfn,然后輸入
examples\NICS_scan\benzene.pdb
25 //離域化和芳香性分析
13 //一維NICS掃描和積分
2 //通過一批原子定義環,將掃描的兩個端點置于環中心上方和下方一定距離處,且連線垂直穿越環中心
1-6 //用于1-6號碳原子定義環。之后屏幕上會顯示通過最小二乘法擬合得到的環平面的法矢量
[回車] //用環上的原子的幾何中心作為環中心。此處也可以自己輸入其它方式得到的中心坐標,比如按照http://www.shanxitv.org/108用Multiwfn做電子密度拓撲分析得到的環臨界點(rcp)
10 //一個端點位于環下方10埃處
10 //另一個端點位于環上方10埃處。之后會從屏幕上看到掃描的起點和終點分別為0,0,-10和0,0,10
200 //掃描的點數。此例相當于每隔約0.1埃一個點,足夠精細了。點數越多計算越耗時
1 //產生Gaussian輸入文件
examples\NICS_scan\template_NMR.gjf //這是一個Gaussian做NMR計算的模板文件。打開它便知計算是在B3LYP/6-31+G*下進行的,和普通輸入文件唯一的差異是坐標部分用[geometry]代替
現在Multiwfn在當前目錄下產生了NICS_1D.gjf,可以用GaussView觀看一下,如下所示,各個紫色的Bq原子是要計算NICS的各個位置(當前原子間的成鍵沒顯示出來,想顯示的話,先在File - Preference - Custom Bonding Parameters里加入Bq與C之間的成鍵判斷設置,將所有類型鍵的判斷閾值都改為0。然后再選Edit - rebond判斷成鍵即可)
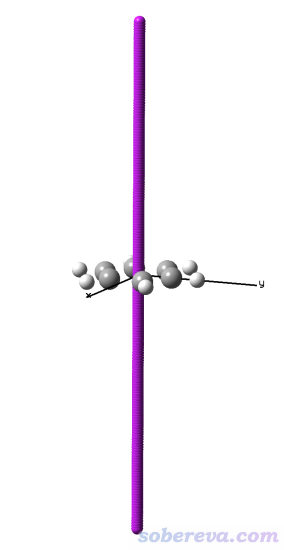
使用Gaussian對NICS_1D.gjf進行計算,我跑好的文件是examples\NICS_scan\benzene_NICS_1D.out。有個不爛的機子話,此任務轉眼就能算完。
接著在Multiwfn的窗口里輸入2,然后輸入benzene_NICS_1D.out的路徑,Multiwfn就會把其中的各個Bq的磁屏蔽張量都載入了。在之后的界面里可以選1和2分別繪制和保存NICS曲線圖像,選3可以導出數據以便在Origin等程序里更靈活地繪制(導出的文本文件里每一列是什么含義在屏幕上提示得超級清楚,別無視),選4可以計算NICS曲線的積分值,選5可以給出曲線的極小和極大點位置和數值。選-1可以設置考察NICS的哪個分量(更確切來說,是指取各個掃描點的磁屏蔽張量的什么分量),一定要記得默認是考察垂直于掃描路徑上的分量,而用戶也可以改成考察自定義矢量上的分量、X或Y或Z分量、各向同性值、各向異性值。
這里選1繪制NICS曲線圖,看到下圖。圖中X=0的位置是環中心位置。可見在分子上方和下方大約1埃處對垂直于環平面方向的磁場的屏蔽最強。
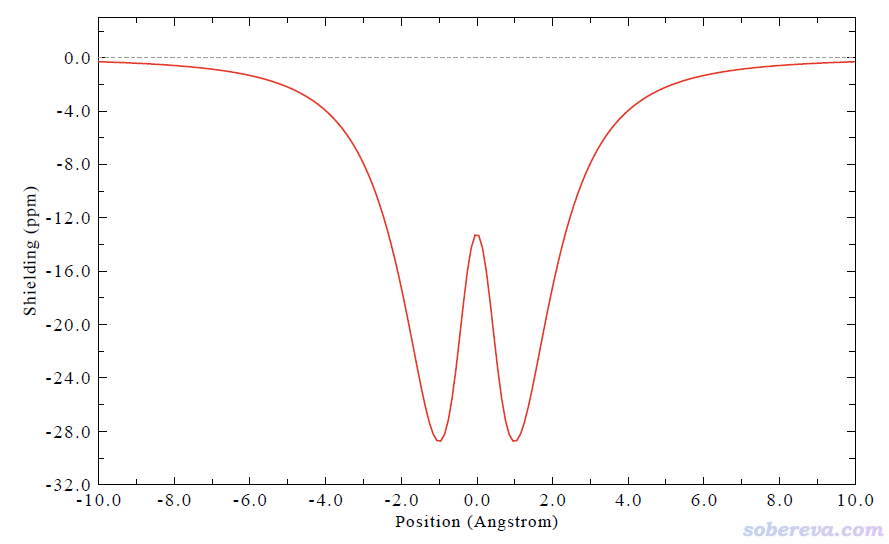
同時在文本窗口也自動顯示了這個NICS曲線的積分值,為-140.78 ppm*Angstrom。由于從上圖看到曲線兩端的數值已經幾乎為0了,所以當前的積分值是準確的。通常掃平面上下、方10埃的范圍就夠了。如果體系兩側是對稱的,只掃描單方向也可以,相應地積分值也只有一半。J. Phys. Chem. A, 123, 3922 (2019)里算的NICS曲線積分值是對于單側掃描情況而言的。
如果在Multiwfn當前界面里選選項5,會輸出如下信息,說明在環平面上方和下方0.955埃處是NICS最小值位置,數值為-28.7 ppm。
Minimum X (Angstrom): -0.954774 Value: -0.28713400E+02
Minimum X (Angstrom): 0.954774 Value: -0.28713400E+02
Totally found 2 minima, 0 maxima
下面再以類似方式對C5H5-和C7H7+計算一維NICS曲線。這倆物質都是和苯分子一樣是6個pi電子的Huckel芳香性體系,但畢竟結構不同,芳香性會存在一定差異,這可以通過對比NICS曲線來考察。此二者的pdb文件、NICS掃描的輸入和輸出文件見examples\NICS_scan目錄下的C5H5-和C7H7+開頭的文件。按照上文做法產生它們的NICS掃描的輸入文件后,在Gaussian計算之前別忘了修改里面的凈電荷設置成為實際情況。
將苯、C5H5-和C7H7+的NICS曲線數據導出然后在Origin里繪制到一起,得到下圖。為了便于展現差異,只顯示了距離平面5埃范圍內的部分。
NICS積分值為C5H5- = -152.70 ppm*Angstrom,C7H7+ = -142.06 ppm*Angstrom。可見單從NICS掃描的角度來看,C5H5-的芳香性比苯和C7H7+都要強。這個結論其實并不準確,因為苯、C5H5-、C7H7+都是pi芳香性體系,但對于芳香性無貢獻的sigma電子對距離平面較近處的NICS會產生一定影響,所以只有單獨考察pi電子對NICS的貢獻才能最嚴格地對比。下一節就演示只考察pi電子貢獻的NICS曲線的繪制。
3 例2:計算垂直于苯環的NICS-sigma和NICS-pi曲線
在《基于Gaussian的NMR=CSGT任務得到各個軌道對NICS貢獻的方法》(http://www.shanxitv.org/670)中我提到,可以利用Gaussian的AICD接口來實現只考察某些軌道對磁屏蔽值貢獻的方法。對于繪制苯的pi電子貢獻的NICS曲線,只需要把上一節例子里的模板文件改為以下內容即可。此文件是examples\NICS_scan\template_NMR_benzene-pi.gjf。
#p b3lyp/6-31+G* nmr=csgt iop(10/93=2)
[空行]
template file
[空行]
0 1
[geometry]
[空行]
AICD.txt
[空行]
17,20,21
[空行]
[空行]
其中nmr=csgt iop(10/93=2)必須寫,AICD.txt是產生的給AICD程序用的文件,當前情況不用管,文件名隨意,產生后可以刪掉。17,20,21是苯的當前的結構在B3LYP/6-31+G*級別波函數的pi軌道序號。想找軌道序號的話,可以在當前級別計算個單點任務得到fch文件,然后根據《使用Multiwfn觀看分子軌道》(http://www.shanxitv.org/269)介紹的Multiwfn主功能0自行挨個觀看軌道圖形判斷,從HOMO開始依次查看序號更小的軌道,直到找到期望個數的pi軌道。也可以根據《在Multiwfn中單獨考察pi電子結構特征》(http://www.shanxitv.org/432)里的做法讓Multiwfn自動判斷(注意判斷pi型分子軌道僅限于純平面體系,此文明確說了)。還要注意一點,NMR=CSGT關鍵詞要求Gaussian以CSGT方式計算磁屏蔽張量,和直接寫NMR關鍵詞默認用的GIAO方式的結果有一定定量差異,但定性一致。比如計算苯的NICS(1)ZZ,用CSGT算的是-27.13 ppm,GIAO算的是-28.76 ppm。所以兩種方式算的結果不適合直接橫向對比。
基于以上模板文件由Multiwfn產生的只考慮pi軌道的輸入文件是examples\NICS_scan\benzene-pi_NICS_1D.gjf,Gaussian計算產生的同名的out文件也在此目錄下提供了。注意當Bq多的時候,nmr=csgt iop(10/93=2)任務的計算耗時會增加非常多。不帶Bq的時候在筆者的2*7R32機子上96核并行4秒鐘算完,而帶了當前200個Bq之后花了39秒。
在Multiwfn里載入NICS掃描輸出文件的時候載入examples\NICS_scan\benzene-pi_NICS_1D.out,然后繪制NICS曲線圖,如下所示。可見在上一節的圖中看到的分子平面處的局部極大點沒了,說明那個極大點是sigma軌道所致,當只考慮pi軌道貢獻的話NICS曲線處處為負。
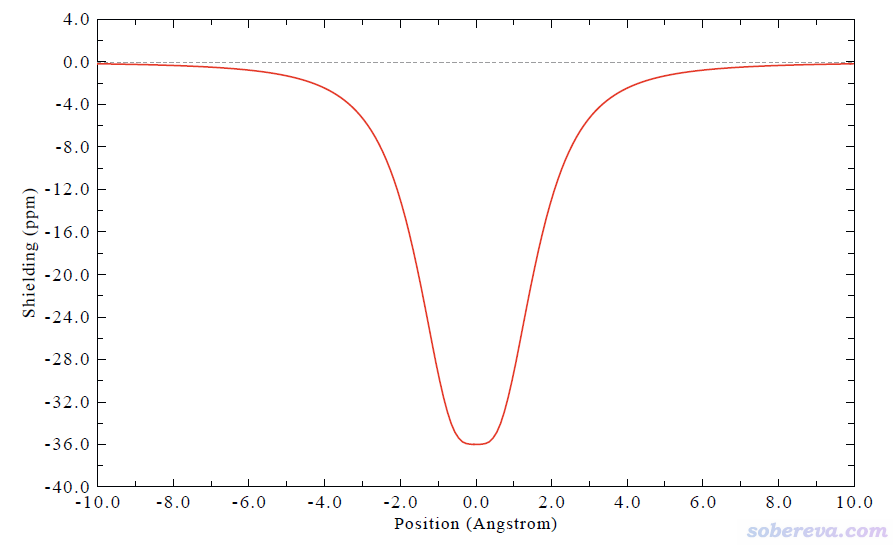
以類似方式,再得到sigma軌道貢獻的NICS曲線,使用的模板文件里軌道序號部分寫1-16,18-19。并且和上面的pi貢獻曲線繪制到一起便于比較,如下所示。可見sigma電子對距離平面很近區域貢獻明顯為正,這充分體現出用NICS(0)ZZ討論pi芳香性體系的芳香性非常不適合,sigma電子的影響會嚴重摻和進去。
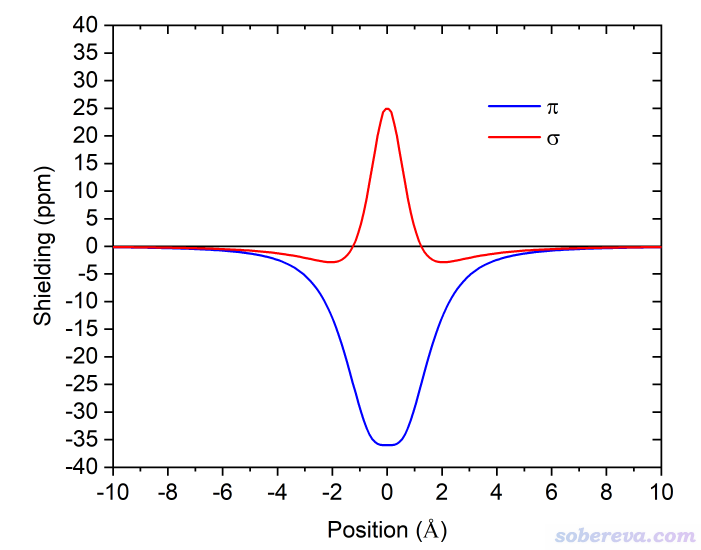
以同樣的方式,對C5H5-和C7H7+獲得sigma和pi電子貢獻的NICS曲線,和苯的曲線繪制在一起便于對比,如下所示
從上圖來看,這三個體系的pi芳香性相仿佛。為了能夠更準確地對比,可以看Multiwfn輸出的NICS曲線積分值,如下所示。可見pi芳香性是C7H7+ >= benzene > C5H5-。
benzene sigma: 14.82 pi: -142.45
C5H5- sigma: -0.38 pi: -134.85
C7H7+ sigma: 14.64 pi: -145.02
值得一提的是,本文計算的結果和NICS曲線積分原文J. Phys. Chem. A, 123, 3922 (2019)里的很一致。以苯的NICS-pi曲線積分值作為100%的話,C7H7+的是101.8%,C5H5-的是94.7%,文獻里是102.3%和94.7%,而文獻里是基于GIAO-B3LYP/6-311+G(d)靠Gaussian結合NBO 7.0做的分解(此文獻表3里的絕對數值和本文的不同,一方面是計算級別不同,一方面是文獻里是從0到∞積分,而本文是[-10,10]埃范圍積分。另外,文獻里依靠NBO 7.0實現分解相對于本文做法的一個缺點是需要花錢購買NBO 7.0)。
以上的pi芳香性的結論通過其它方式也可以證明。比如用Multiwfn做ELF-pi的拓撲分析得到二分點數值,C7H7+為0.936,苯為0.897,C5H5-為0.803,也是C7H7+ >= benzene > C5H5-的順序。如果你不了解怎么做這種分析的話,看《在Multiwfn中單獨考察pi電子結構特征》(http://www.shanxitv.org/432)。
4 例3:計算垂直于無窮烯中某苯環的NICS曲線
在《利用Multiwfn計算傾斜、扭曲環的NICS_ZZ》(http://www.shanxitv.org/261)中介紹過Multiwfn可以對扭曲、傾斜的環計算NICS_ZZ值,Multiwfn計算一維曲線的功能也同樣可以考慮這樣的環。這里以無窮烯中間的環作為例子,結構文件是examples\NICS_scan\infinitene.pdb,是在PBE0/6-31G(d)級別優化過的,如下所示,給了兩個不同的視角。此例要研究的是高亮的原子構成的環,這部分的環明顯是斜著的而且是略微扭曲的。
由上圖可見,不可能在被高亮的環上方和下方對稱地進行掃描,否則其中一頭掃描的點就會接觸到另一側的環了。因此,本例只從這個環的中心垂直向外側掃描10埃距離。
啟動Multiwfn,然后輸入
examples\NICS_scan\infinitene.pdb
25 //離域化和芳香性分析
13 //一維NICS掃描和積分
2 //通過一批原子定義環,將掃描的兩個端點置于環中心上方和下方一定距離處,且連線垂直穿越環中心
35-37,68-69,71 //用于35-37,68-69,71號碳原子定義環,即上圖高亮的那些原子
[回車] //用環上的原子的幾何中心作為環中心
10 //一個端點位于環中心下方10埃處
0 //另一個端點位于環上方0埃處,也即環中心處
[回車] //掃描的點數用推薦的100(推薦值是以間隔0.1埃確定的)
1 //產生Gaussian輸入文件
examples\NICS_scan\template_NMR.gjf //Gaussian做NMR計算的模板文件
用GaussView觀看在當前目錄下產生的NICS_1D.gjf,如下所示。可見Bq確實出現在了期望的位置。如果對某個體系你發現Bq分布在了與預期相反方向,就把環上方和環下方的距離反過來輸入重新產生輸入文件即可。
為節約時間,把NICS_1D.gjf里的6-31+G*改為6-31G*,然后用Gaussian計算之,得到的文件是examples\NICS_scan\infinitene_NICS_1D.out。之后在Multiwfn里輸入2,輸入此文件路徑,再選1繪圖,看到下面的NICS曲線圖(如果想讓此圖左右翻轉,可以選選項-3)
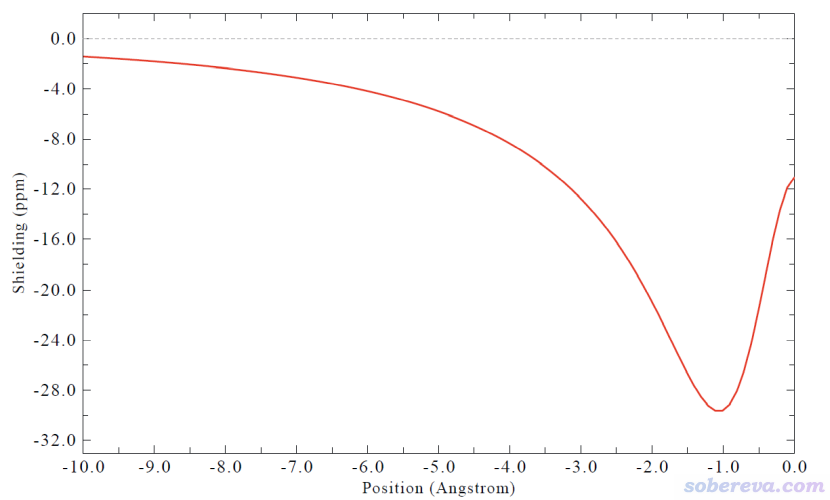
此曲線積分值為-96.46 ppm*Angstrom。僅對環的單側進行積分數值就已經很大了,明顯大于第2節苯分子的兩側NICS積分值的一半(約-70 ppm)。其原因絕非是因為無窮烯中這個環的芳香性比苯還強,而是附近其它的環產生的磁屏蔽效果產生了疊加。使用Multiwfn計算《衡量芳香性的方法以及在Multiwfn中的計算》(http://www.shanxitv.org/176)里介紹的多中心鍵級或者《使用Multiwfn計算AV1245指數研究大環的芳香性》(http://www.shanxitv.org/519)里介紹的AV1245分析,就可以明顯看到這個環的芳香性并不如苯。
順帶一提,是否帶彌散函數對NICS曲線影響甚微,不帶彌散函數的話耗時能巨幅降低,尤其是對于無窮烯這樣較大的體系。6-31G*和6-31+G*在2*7R32 96核機子上計算當前任務分別耗時1m6s和4m37s。6-31G*和6-31+G*算的NICS積分值分別是-96.46和-96.19 ppm*Angstrom,相差微乎其微,畫出圖來對比也幾乎沒可察覺的差異。之前的例子帶了彌散函數主要是考慮到要將C5H5-陰離子體系納入對比。只是算中性體系的NICS沒太大必要帶彌散,哪怕要掃描到較遠處。想要比6-31G*更好的結果可以用3-zeta檔次的基組。
5 總結
本文介紹了Multiwfn掃描一維NICS曲線的功能,既可以直接作圖又可以給出積分值,比只討論環中心或上/下方1埃處的值獲得的信息更充分,討論芳香性時更嚴格,而且還能分解成不同軌道的貢獻。從例子可見這種分析在Multiwfn里的操作非常簡單。為了確保計算合理,建議在Multiwfn產生NICS掃描的gjf文件后放到GaussView里檢查一下Bq的位置是否確實正確,如果和預期的不符說明在Multiwfn里輸入的信息有誤。
使用Multiwfn此功能發表文章請記得按Multiwfn啟動后顯示的信息恰當引用Multiwfn的原文。
]]>文/Sobereva@北京科音 2023-Jun-4
0 前言
平均局部離子化能(average local ionization energy, ALIE)是波函數分析領域里很常用的實空間函數,它衡量三維空間中特定位置的電子發生電離的難易程度,數值越小體現電子被體系束縛得越弱、越容易發生親電反應(被親電進攻)。ALIE也體現局部的親核性。ALIE的分析可以使用波函數分析程序Multiwfn(http://www.shanxitv.org/multiwfn)非常容易地實現,相關介紹見《使用Multiwfn的定量分子表面分析功能預測反應位點、分析分子間相互作用》(http://www.shanxitv.org/159)、《使用Multiwfn和VMD繪制平均局部離子化能(ALIE)著色的分子表面圖(含視頻演示)》(http://www.shanxitv.org/514)、《親電取代反應中活性位點預測方法的比較》(http://www.whxb.pku.edu.cn/CN/abstract/abstract28694.shtml),以及《靜電勢與平均局部離子化能相關資料合集》(http://bbs.keinsci.com/thread-219-1-1.html)中的相關綜述。
ALIE可以用來預測和解釋親電反應的區域選擇性和快慢,由于它很有用,而且通過Multiwfn實現分析特別容易,使得其如今已經很流行。是否有與之用途類似的函數能夠考察親核反應?答案是Y。有兩個函數專門衡量局部的親電性,都能夠拿來討論化學體系發生親核反應的位點和反應速率,且和ALIE一樣都是基于分子軌道波函數和軌道能級定義的。其中一個是J. Mol. Model., 9, 342 (2003)中提出的local electron affinity(局部電子親和能,LEA),另一個是J. Phys. Chem. A., 120, 10023 (2016)中提出的local electron attachment energy(局部電子附著能,LEAE)。相對來說LEAE更好一些,在于LEA計算時依賴于所有空軌道,導致它的基組敏感性很強,特別是基組帶彌散函數時得到的結果往往沒有意義。而LEAE計算時只依賴于能量低于0的空軌道,對基組敏感性小得多,更皮實,但必須保證至少LUMO的能量是低于0的。LEAE原文建議使用B3LYP/6-31+G**產生的波函數做LEAE計算,此時總能保證這一點。本文只介紹LEAE,如果讀者對LEA感興趣,參看Multiwfn手冊4.12.13節的介紹和例子。
下文第1節先介紹LEAE的定義和實用價值,第2節將介紹怎么用Multiwfn給出分子表面上LEAE極小點的位置和具體數值,這是定量分析的關鍵。第3節介紹怎么利用腳本便利地實現Multiwfn結合VMD繪制分子表面的LEAE著色圖,這對于通過圖像直觀討論非常重要。
1 局部電子附著能的概念
1.1 局部電子附著能的定義
LEAE可表示為下式
其中ρ是總電子密度,|φ_i|^2是分子軌道i的概率密度,ε是軌道能量。循環考慮從LUMO開始的所有能量低于0的空軌道。對限制性閉殼層波函數,n為2,對非限制性開殼層波函數,n為1。在Multiwfn中LEAE對應于第-27號自定義函數,可以使用Multiwfn對其像對其它實空間函數一樣做各種形式的分析、繪圖。
LEAE較負的地方,說明在此處有能級較負的空軌道出現,也因此這個地方比較親電,即傾向于與帶負電的物質相結合,同時也說明這種地方傾向于被親核進攻。當同一個分子有多個LEAE較負的地方,誰越負誰越親電、被親核進攻的優先級越高(忽略位阻、溶劑效應等外部因素)。LEAE還可以在不同體系之間對比,即對于一批類似分子,發生反應的位點的局部分子表面區域的LEAE越負,發生親核反應的速率常數通常越大。
通常LEAE是像ALIE那樣投影到分子表面上考察的。可以用類似《使用Multiwfn+VMD快速地繪制靜電勢著色的分子范德華表面圖和分子間穿透圖(含視頻演示)》(http://www.shanxitv.org/443)和《基于Multiwfn產生的cube文件在VMD和GaussView中繪制填色等值面圖的方法》(http://www.shanxitv.org/402)所述的方式繪制分子表面LEAE著色圖來一目了然地展現不同位置的LEAE大小。也可以使用比如J. Mol. Graph. Model., 38, 314 (2012)中我提出的定量分子表面分析算法尋找LEAE在分子表面上的極小點,根據它們大致對應的原子以及具體數值討論親核反應。還可以用我在Multiwfn中獨家實現的“局部分子表面分析”計算各個暴露的原子在分子表面上的局部區域中的LEAE的平均值討論,這樣即便某個原子附近沒有LEAE的表面極小點出現也照樣能用LEAE來分析,參考下文以及《談談怎么計算“原子的靜電勢”》(http://www.shanxitv.org/641)中與局部分子表面分析有關的介紹。
特別要注意的是,雖然靜電勢、ALIE通常都是投影到電子密度為0.001 a.u.的等值面(Bader定義的氣態分子的范德華表面)上進行分析的,但LEAE原文里作者建議將LEAE投影到0.004 a.u.等值面上進行分析,結果更好。因此后文的例子都遵循原文的這個做法。
討論親核反應的方法還有很多,比如LUMO分布、福井函數f+、軌道權重福井函數fw+、Hirshfeld原子電荷、分子表面靜電勢等,《預測親核反應位點方法的比較》(https://link.springer.com/article/10.1007%2Fs11426-015-5494-7)里面有很多介紹和對比。這里說說LEAE和它們的關系:
? 相對于靠前線軌道理論根據LUMO分布來預測親核反應位點,LEAE的好處在于考慮的不僅僅是LUMO。當空軌道能級非常接近(近簡并)的情況,只拿LUMO說事會有嚴重誤導,而LEAE則沒有這個問題。
? 相對于福井函數f+,LEAE的一個好處是只需要對一個電子態進行計算,而不用算兩個電子態再將其密度求差,因而更省事(不過按照《使用Multiwfn超級方便地計算出概念密度泛函理論中定義的各種量》http://www.shanxitv.org/484說的用Multiwfn來計算f+也極其簡單),而且可以在不同體系間對比。
? 《通過軌道權重福井函數和軌道權重雙描述符預測親核和親電反應位點》(http://www.shanxitv.org/533)中介紹的軌道權重福井函數當中的fw+與LEAE有較大相似性,即都基于非占據分子軌道計算且又不止考慮LUMO,而二者的思想又有所不同。fw+是基于人為設定的權重函數決定最低一批空軌道各自的權重,而LEAE中各空軌道的權重隱含地體現在了軌道能量里。fw+和LEAE沒法說誰更好,畢竟考察方式不一樣:fw+通常繪制等值面圖考察,而且由于是歸一化的函數,可以討論各個原子的貢獻百分比,而LEAE通常是對比它在分子表面上不同區域的數值。LEAE比fw+有一個明確的優點是它可以直接在不同分子間對比絕對大小,不限于fw+和f+那樣只能在單個分子內對比。
? 在《TCA上的一篇對比不同原子電荷預測反應位點、親電/親核性的文章》(http://bbs.keinsci.com/thread-15512-1-1.html)里我提到過Hirshfeld原子電荷是討論親核位點和反應速率的很有用的量,《預測親核反應位點方法的比較》(https://link.springer.com/article/10.1007%2Fs11426-015-5494-7)的測試也體現了這一點。LEAE和Hirshfeld電荷在這方面誰更好不一定,我還沒見過全面的對比測試,在我來看二者都可以同時使用來相互印證。Hirshfeld電荷是一個原子一個值,雖然在討論原子的特征上顯得比LEAE更方便,但它沒法像LEAE那樣考察特定局部區域的親電性(如化學鍵、sigma穴、pi穴區域等),而且沒法畫成LEAE那樣直觀的分子表面著色圖(頂多是按照《使用Multiwfn+VMD以原子著色方式表現原子電荷、自旋布居、電荷轉移、簡縮福井函數》http://www.shanxitv.org/425的做法對原子著色來圖形化展現),所以二者有互補性。
? 靜電勢在討論親核反應上有一定用處,分子表面上靜電勢越正的地方被認為越親電、越容易發生親核反應。LEAE和靜電勢在考察形式上相近。但如LEAE下文中所展現的,在分析親核反應方面靜電勢遠不如LEAE可靠。
1.2 局部電子附著能在研究親核反應上的用處
LEAE原文給了不少例子,這里挑幾個說說。
下圖a是五氟硝基苯的分子表面(對于LEAE分析來說指0.004 a.u.電子密度等值面,后同),其中最負的部分(< -2.05 eV)的區域為紅色。可以看到硝基臨位和對位各有一塊紅色,其中對位碳的表面LEAE極小值為-2.21 eV,臨位為?2.11 eV。這體現出五氟硝基苯傾向于在臨位和對位發生親核反應。確實,實驗上此體系是選擇性地在臨、對位發生親核芳香取代反應的。在圖上硝基的氮上也能看到一丟丟紅色區域,相應的表面LEAE極小點數值為-2.06 eV,說明N的pi區域有親電性(所謂的pi穴)。
上圖b是五氟硝基苯的LUMO等值面圖,可見LUMO在間位的碳上也有非常明顯的分布,因此LUMO對此體系的鄰、對位發生選擇性親核反應的特點表現得不夠充分,遠不如LEAE。上圖c是表面靜電勢圖,紅色是最正的部分(>25 kcal/mol),根本都沒體現出鄰、對位的碳在發生親核反應時比間位的碳有任何優勢,因此對預測親核位點完全失敗。之所以苯環正上方區域靜電勢為正,這是因為當前苯環連的都是吸電子基團,導致苯環pi電子密度較低,對核電荷對靜電勢的正貢獻抵消減弱所致;另一方面,根據計算公式可知,靜電勢是有跨空間效應的,某個碳的pi電子的減少會影響到附近區域的靜電勢,各個碳的這個效應疊加導致靜電勢最正的區域出現在了它們的中央。
下圖是一系列氟取代的雜環體系,LEAE都正確預測對了發生親核芳香取代的位點,并且圖中把相應位點的表面LEAE極小值與反應速率常數k的ln之間的關系繪制了出來,可見有很好的線性相關性,即反應位點處分子表面LEAE越負,發生親核反應的速率常數越大。
LEAE原文考察了不同方式取代的硝基二苯乙烯,如下圖所示,發現表面LEAE極小值出現的位置都在β碳上,正對應于HOCH2CH2S-與它們發生親核加成的位置。并且這些極小值與ln(k)有極好的線性關系。這再次體現出LEAE在判斷親核反應位點和難易方面很有用。一旦擬合出這種關系后,R3、R4為其它基團時的k就能較準確且很方便地預測出來。
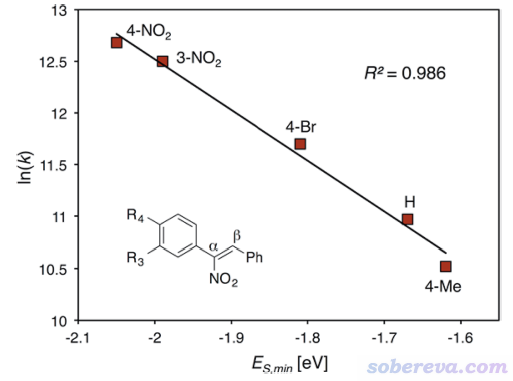
1.3 局部電子附著能在研究弱相互作用上的用處
下圖左側是溴代甲烷分子表面的LEAE,最負的地方是紅色,較負的地方是黃色,可見LEAE一方面展現了Br的顯Lewis酸性的sigma-hole區域,一方面把CH3部分能被SN2親核進攻的地方展現了出來。順帶一提,親核進攻方向還可以用《通過電子定域化函數(ELF)、價層電子密度分析討論親核進攻的方向》(http://www.shanxitv.org/606)介紹的方法討論。
靜電勢在討論靜電效應對分子間相互作用的控制方面用得極其普遍,例如《靜電效應主導了氫氣、氮氣二聚體的構型》(http://www.shanxitv.org/209)、《全面探究18碳環獨特的分子間相互作用與pi-pi堆積特征》(http://www.shanxitv.org/572)。上圖右側是分子表面靜電勢,由最負到最正按照藍-綠-黃-紅變化,可見靜電勢和LEAE反應的信息既有共性也有差異。靜電勢除了把Br的sigma-hole展現出了以外,還把Br原子的一圈孤對電子導致的Lewis堿性區域(藍色部分)展現了出來。在CH3一側,靜電勢較正的區域除了能受到SN2進攻的部分外,還有帶正電的氫的部分。因此相對于靜電勢比較完整地展現分子表面各區域的親核親電特征來說,LEAE僅展現出親電部分,而且還僅限比較“軟”(易發生電子轉移和極化)的親電部分。因此在研究弱相互作用方面,LEAE與靜電勢有明顯互補性。
下圖是不同的鹵代甲烷形成鹵鍵的位點的表面LEAE極小值和相互作用能之間的關系,可見線性相關性極好。這充分體現出LEAE不僅能用來討論親核反應,在研究弱鹵鍵作用方面也頗有價值(用于磷鍵、碳鍵等其它以hole作為Lewis酸形成的靜電主導的相互作用方面應當也有類似的價值)。
LEAE原文發現鹵原子表面的LEAE極小值與靜電勢極大值有較高相關性,但也有一定互補性,如果將二者同時作為變量構建與相互作用能的關系的話,可以得到更理想的線性關系。下圖的鹵代苯形成的鹵鍵相互作用能就是例子,紅線標注的式子里E_S,min是鹵原子表面LEAE極小點數值,V_S,max是其表面靜電勢極大點數值。
2 使用Multiwfn計算分子表面上局部電子附著能的極值點
這一節就以五氟硝基苯(C6F5NO2)為例具體介紹如何通過Multiwfn計算分子表面上LEAE極值點位置和數值,其實操作和《使用Multiwfn的定量分子表面分析功能預測反應位點、分析分子間相互作用》(http://www.shanxitv.org/159)里介紹的非常相似。本文例子涉及的所有文件在http://www.shanxitv.org/attach/676/file.zip里都有。坐標都是用LEAE原文里用的B3LYP/6-31G*級別優化的,.fch波函數文件都是Gaussian 16在B3LYP/6-31+G**下算單點產生的。
讀者一定要使用2023-Jul-4及以后更新的Multiwfn。Multiwfn可以在官網http://www.shanxitv.org/multiwfn免費下載。對Multiwfn不了解者參見《Multiwfn入門tips》(http://www.shanxitv.org/167)、《Multiwfn FAQ》(http://www.shanxitv.org/452)、量子化學波函數分析與Multiwfn程序培訓班(http://www.keinsci.com/workshop/WFN_content.html)。如果你還不知道怎么產生Multiwfn做波函數分析所需的輸入文件如.fch,看《詳談Multiwfn支持的輸入文件類型、產生方法以及相互轉換》(http://www.shanxitv.org/379)。注意,計算LEAE所用的波函數文件必須帶有空軌道信息,因此不能用比如.wfn這種不含空軌道信息的格式當Multiwfn的輸入文件。
本文文件包里的C6F5NO2.gjf是在B3LYP/6-31G*優化的結構上做B3LYP/6-31+G**計算的Gaussian輸入文件,用Gaussian運行之,得到C6F5NO2.chk。然后用formchk將之轉成fch格式,所得的C6F5NO2.fch在本文的文件包里已經提供了。啟動Multiwfn,載入此文件,然后輸入
12 //定量分子表面分析
2 //選擇映射的函數
-4 //LEAE(如果選4則是LEA)。注意之后Multiwfn自動把分子表面的定義從默認的0.001 a.u.電子密度等值面切換為了0.004 a.u.
0 //開始分析
電子密度和LEAE計算耗時極低,當前體系又小,Multiwfn的效率又非常高,因此一瞬間就算完了。注意屏幕上顯示了分子表面上LEAE的各種統計量,以及所有極小點和極大點坐標和數值。我們感興趣的只有表面極小點,結果列在了下面,以各種單位表示的LEAE值都給出了,帶星號的是表面最小點數值
The number of surface minima: 23
# a.u. eV kcal/mol X/Y/Z coordinate(Angstrom)
1 -0.06045870 -1.645165 -37.938437 -2.053417 -0.331424 -3.315850
2 -0.06891843 -1.875366 -43.247002 -1.507986 1.481935 1.357405
3 -0.07754516 -2.110111 -48.660361 -1.469940 -1.570369 -0.320762
4 -0.06736260 -1.833030 -42.270707 -1.470715 -1.465427 1.483792
* 5 -0.08159589 -2.220337 -51.202240 -1.484711 0.007741 2.216138
6 -0.07346095 -1.998974 -46.097480 -1.500188 1.473500 -0.336122
7 -0.07536621 -2.050819 -47.293049 -1.074943 0.940426 -1.922978
8 -0.01117322 -0.304039 -7.011307 -0.827293 3.377774 -1.353531
9 -0.01055470 -0.287208 -6.623180 -0.614413 -3.455994 -1.378276
10 -0.01339108 -0.364390 -8.403035 -0.039211 -3.526215 2.507591
11 -0.06047453 -1.645596 -37.948370 -0.024936 -2.058019 -3.319430
12 -0.01491144 -0.405761 -9.357077 -0.017163 -0.002495 4.539262
13 -0.06052520 -1.646974 -37.980166 0.048059 2.074908 -3.302895
14 -0.01339150 -0.364401 -8.403302 0.058848 3.544428 2.473038
15 -0.01054264 -0.286880 -6.615611 0.589544 3.454318 -1.406822
16 -0.01117424 -0.304067 -7.011948 0.868346 -3.349096 -1.351258
17 -0.07520552 -2.046446 -47.192218 1.155106 -0.842080 -1.924120
18 -0.06890413 -1.874977 -43.238030 1.510073 -1.490657 1.329452
19 -0.07346343 -1.999042 -46.099037 1.508220 -1.439197 -0.339503
20 -0.08158888 -2.220146 -51.197836 1.484706 0.011734 2.216151
21 -0.06734033 -1.832423 -42.256728 1.467340 1.488012 1.473255
22 -0.07751511 -2.109293 -48.641507 1.473641 1.590240 -0.223002
23 -0.06050316 -1.646375 -37.966336 2.049444 0.308546 -3.302895
在后處理菜單里選擇0,會進入觀看結構和極值點的圖形界面。將窗口右側的Ratio of atomic size設大到3.0,取消選擇Maximum position復選框來隱藏極大點,然后選中Minimum label復選框以顯示極小點序號,通過Size of labels適當調整標簽的大小,此時看到的圖像如下所示
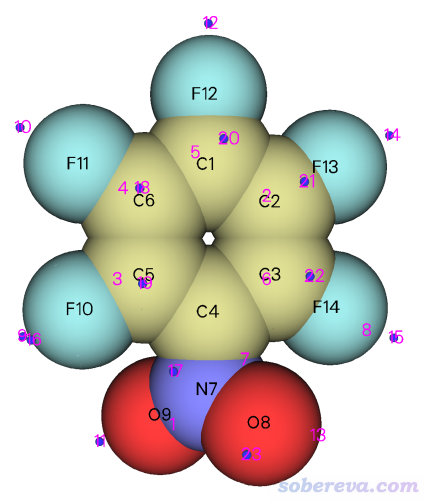
上圖中藍色小球是表面LEAE極小點位置,可見在鄰、間、對位碳的上方都有極小點。對照文本窗口顯示的數值,可知對位碳的表面LEAE極小點(20號)的值是-2.22 eV,和LEAE原文里給出的幾乎精確一致。兩個臨位碳的極小點是19和22號,數值分別為-2.00和-2.11 eV,實際討論時可以取平均值。在兩個間位碳上也有極小點,是18和21號,數值分別為-1.87和-1.83 eV,明顯比臨、對位的更正,體現出間位不是親核反應發生的優先位點。17號極小點與氮相對應,這也正體現前述的氮的pi-hole的存在。
這里也順便對LEAE做一下我提出的局部分子表面分析,具體介紹可以看Multiwfn手冊3.15.2.2節,這比起僅僅考察極值點一個位置的數值更能全面地展現原子的特征。在Multiwfn后處理菜單選擇11,馬上看到各個原子局部表面上的LEAE的統計量,其中一部分如下所示,這體現的是各個原子局部表面上LEAE所有值/正值部分/負值部分的平均值(由于沒有負值部分,所以這部分為NaN,即not a number)。可見對位碳(1號)局部表面上LEAE平均值為-1.455 eV,是所有原子最負的,肯定特別容易發生親核反應。臨位碳(3號和5號)其次,約-1.31 eV,而間位碳2和6號原子為-1.29 eV,比鄰、間位更正,故最不容易發生親核反應。此結論和基于表面極小點進行分析是一致的。
Atom# All/Positive/Negative average
1 -1.45512 NaN -1.45512
2 -1.28606 NaN -1.28606
3 -1.31096 NaN -1.31096
4 -1.06736 NaN -1.06736
5 -1.30834 NaN -1.30834
6 -1.28796 NaN -1.28796
7 -0.84143 NaN -0.84143
8 -0.57892 NaN -0.57892
9 -0.58036 NaN -0.58036
10 -0.30322 NaN -0.30322
11 -0.34908 NaN -0.34908
12 -0.39730 NaN -0.39730
13 -0.34974 NaN -0.34974
14 -0.30276 NaN -0.30276
3 使用Multiwfn結合VMD繪制分子表面的局部電子附著能
本節介紹怎么在Windows下基于Multiwfn和VMD方便地繪制漂亮的LEAE著色的分子表面圖。本文用的VMD是1.9.3版,可以在http://www.ks.uiuc.edu/Research/vmd/免費下載。這里利用了Windows批處理文件以簡化操作,閱讀《詳談Multiwfn的命令行方式運行和批量運行的方法》(http://www.shanxitv.org/612)可以充分了解利用批處理文件運行Multiwfn的原理。
先進行準備工作:將Multiwfn目錄下的examples\scripts\local_EA子目錄下的LEAE_isoext.txt和LEAE_isoext.bat都拷到Multiwfn可執行文件所在目錄,將LEAE_isoext.vmd拷到VMD目錄下(即VMD啟動后在文本窗口輸入pwd命令顯示的目錄)。
此例繪制1-溴-3,5-二氟苯。其波函數文件是本文file文件包里的C6F2H3Br.fch,產生此文件用的Gaussian輸入文件C6F2H3Br.gjf也一起給出了。進行以下操作:
(1)C6F2H3Br.fch拷到Multiwfn可執行文件所在目錄
(2)在LEAE_isoext.bat上點右鍵,選“編輯”,第一行Multiwfn后面的文件名改成C6F2H3Br.fch。然后把此文件里2、3、4行的VMD目錄改成你機子里VMD的實際目錄,之后保存文件
(3)雙擊LEAE_isoext.bat批處理文件運行。此時它會把LEAE_isoext.txt里記錄的命令傳遞給Multiwfn進行運算,產生的userfunc.cub、density.cub、surfanalysis.pdb會自動被挪到VMD目錄下
(4)啟動VMD,輸入source LEAE_isoext.vmd來運行作圖腳本,然后馬上看到下圖
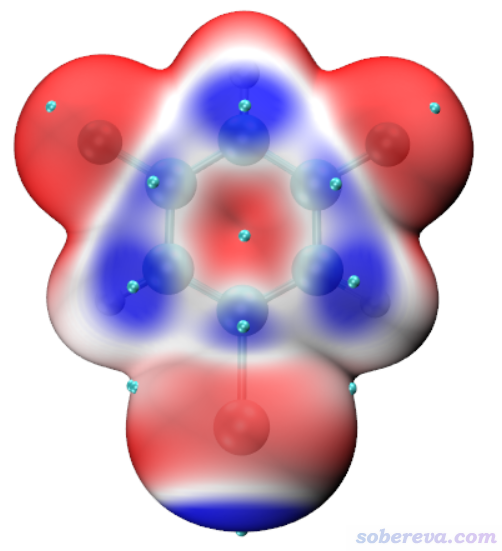
此圖展現了電子密度=0.004 a.u.對應的分子表面的LEAE。腳本默認的色彩刻度是-0.03到0.0 eV按照藍-白-紅變化,即越藍的地方LEAE越負、親電性越強、越易發生親核反應。從此圖可以清楚地看出溴的sigma-hole區域很親電,以及溴的鄰、對位的碳的親電性比間位的碳更強。圖中青色小球是表面極小點位置。
如果在默認的色彩刻度范圍下分子表面只有一種顏色,或者顏色區分不開的話,需要自行調節色彩刻度范圍,比如在VMD文本窗口輸入mol scaleminmax 0 1 -0.04 0.0就可以把色彩刻度改為-0.04到0.0。在LEAE_isoext.vmd腳本里也可以直接改默認的色彩刻度。
關于色彩刻度條的顯示問題,參考《使用Multiwfn+VMD快速地繪制靜電勢著色的分子范德華表面圖和分子間穿透圖(含視頻演示)》(http://www.shanxitv.org/443)里相應部分。
想查詢各個表面極小點數值的話,點擊VMD的圖形窗口激活之,再點擊鍵盤上的0進入VMD的查詢模式(此時光標會變成十字),然后點擊一個表面極小點對應的圓球的正中心,文本窗口里如果顯示比如index 36,就在文本窗口輸入[atomselect top "index 36"] get beta然后回車,此時顯示的數值就是以eV為單位的LEAE值。
按照以上說明,把色彩刻度軸添加、把表面LEAE極小點數值標注,并且把色彩刻度設為-0.04至0.0 eV后,看到的圖像如下所示,可見效果很好。
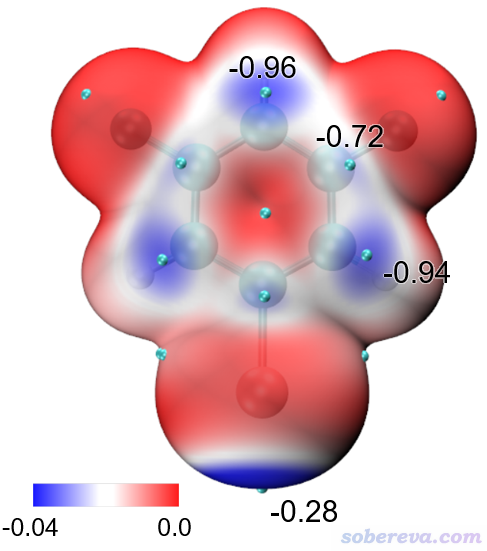
作圖腳本默認是對等值面使用EdgyGlass材質,大家如果想修改顯示效果,可以進入VMD的Graphics - Materials界面,選擇EdgyGlass,然后調節各種材質屬性,比如透明度等。
值得一提的是,此例也反映出LEAE比起只靠LUMO分析親電位點的一個明顯優點。下圖是按照《使用Multiwfn觀看分子軌道》(http://www.shanxitv.org/269)繪制的此體系的LUMO和LUMO+2等值面圖。可見LUMO完全在苯環上,如果只拿LUMO說事,根本體現不出Br的sigma-hole的親電特征,而LUMO+2才把sigma-hole給體現出來。在當前計算級別下,LUMO到LUMO+3的能量都低于0,因而都被LEAE所納入了,這是為什么LEAE能同時展現出sigma-hole和芳環上的親電區域。
本文的文件包里還有個benzylidenemalononitrile.fch,下面是對這個體系作的LEAE圖以及對各個表面極小點標注的LEAE值,色彩刻度用-0.08至0.0 eV。圖中最藍的地方是β碳,正對應于此體系能發生親核反應的位置。取代基的臨、對位碳上LEAE比其它碳更負,也正對應于這倆位置是選擇性發生親核芳香取代的位點。
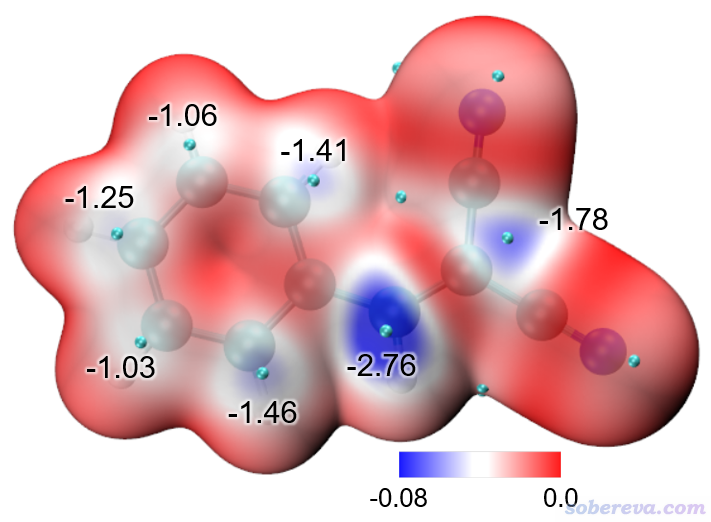
4 總結
本文對局部電子附著能(local electron attachment energy,LEAE)的定義、特點做了介紹,并且討論了它與其它有類似用途的分析方法的異同。從本文的例子可見,LEAE不僅能用于預測/解釋一個分子中的親核反應位點,還能橫向對比類似分子發生同類親核反應的難易,而且還能用于討論鹵鍵、碳鍵、磷鍵等通過局部顯Lewis酸特征的hole形成的弱相互作用。通過Multiwfn,可以非常便利、快速地實現LEAE的分子表面定量分析,還能結合VMD繪制美觀、直觀又很能說明問題的LEAE著色的分子表面圖像。LEAE無疑像平均局部離子化能(ALIE)和靜電勢一樣是非常有實用價值的實空間函數,值得在實際研究中利用。分子表面LEAE極小值也可以視為一種有意義的分子描述符,給《Multiwfn可以計算的分子描述符一覽》(http://www.shanxitv.org/601)介紹的Multiwfn能算的眾多描述符中又增加了新的一員。本文中的這些分析手段對于ALIE、靜電勢同樣適用,前面文中引用的博文里都有詳細說明。
使用Multiwfn做LEAE分析請務必記得按照程序包里的How to cite Multiwfn.pdf中的說明對Multiwfn的原文進行恰當引用。如果涉及到討論分子表面LEAE極值點,也請同時引用J. Mol. Graph. Model., 38, 314 (2012),其中詳細介紹了Multiwfn做定量分子表面分析、搜索表面極值點的算法。
補充:有讀者看完本文后,在使用Multiwfn做LEAE分析時發現沒有結果。要么是輸入文件沒用對(不含波函數信息,或者不含空軌道),要么就是LUMO的能量為正,這不滿足本文明確說的計算LEAE的要求。LUMO能量為正時,確保基組用的是LEAE原文建議用的6-31+G**(或者其它帶彌散函數的基組,如ma-def2-TZVP),并且結合B3LYP或者與之HF成份差不多的泛函如PBE0。如果此時LUMO能量也為正,那就沒法用LEAE了,只能靠別的方式來考察,比如上文提及的福井函數或軌道權重福井函數fw+。
]]>